
Podcast: Metabolic Syndrome Explained
Metabolic syndrome is a collection of risk factors that can ultimately increase the risk of developing a variety of health issues, including heart disease, stroke, and diabetes, among other problems. Central obesity, high blood pressure, high blood sugar, high triglycerides, and low HDL are the 5 risk factors associated with metabolic syndrome. Having at least three of the five risk factors may suggest the presence of metabolic syndrome. Dr. Alex Jimenez and Dr. Mario Ruja explain the 5 risk factors associated with metabolic syndrome, in further detail, as they recommend diet and lifestyle modification advice and guidelines to help people with metabolic syndrome improve their overall health and wellness. From eating fiber and staying hydrated to exercise and better sleep, Dr. Alex Jimenez and Dr. Mario Ruja discuss how diet and lifestyle modifications can help improve the 5 risk factors associated with metabolic syndrome to ultimately prevent the risk of developing a variety of other health issues, including heart disease, stroke, and diabetes. – Podcast Insight
[00:00:07] And we are live. Yes, we are. Hi, this is Dr. Alex Jimenez. Today we’re gonna be talking with Dr. Mario Ruja. We’re here together today. We’re testing out a new technology of head to head conversations regarding the whole process. Mario, how you feeling, baby?
[00:00:24] Feeling incredibly metabolic, Alex.
[00:00:29] Yes, really metabolic. I’m about to go through this mic right now. That’s what I’m talking about. Hey, we’re here.
[00:00:37] Mario and I are, you know, we’re gonna be hitting you every day. Every week. Every time we can. As much as we can. We’re gonna be going through the airways. Yeah. And we’re gonna be using the new technology to discuss exactly what we’re up to. Today, we’re focusing on an interesting disorder called metabolic syndrome. Many of you have heard the word. But really, you know, tying in exactly what it is that we’re talking about requires kind of elaborate conversation. You’ve seen it in many pictures. Mario, you can pop up the picture there PIP and you can see that a lot of times people see this gut thing going on. And that’s one of the components of it. Metabolic syndrome, when you break it down is ultimately and people notice it when they go to their doctors. Doctors are very good at assessing clinical assessments at the point where they show up in the lab work. Now, metabolic syndrome is one of these issues that many people have. And when they’re diabetic, well, they are pretty much in that range already.
[00:01:39] But before it happens, the body can stray into a metabolic area where a lot of times, for example, if your blood sugar is over 100 and you’re starting to feel like really bad, your bellies are really large. We need to have some parameters to determine it. But most people end up having metabolic syndrome and just feeling like crap. So the idea behind this process and understanding what metabolic syndrome is, understanding that there are some underlying pathologies with it. So what we’re going to talk about today is we’re going to talk about issues that are related to it. Now, in the areas of diabetes, we have, you know, complicating issues such as sleep apnea, large waistline, people who take metformin, liver disorders, nonalcoholic delivered diseases that we have heard of all fall under the realm of metabolic. But we have certain criteria that we can do that actually determines what metabolic syndrome is. Now, Mario, you’ve noticed some things regarding blood pressure. Yeah. Now, if you can show the PIP and when we see this, we can actually determine if you can kind of explain that a little bit.
[00:02:44] Yeah, it’s very simple, when you’re looking at blood pressure, your whole system. When you are out of balance in terms of your sugar, Alex, and your gut is overflowing your belt and you have issues tucking in your shirt. Now that blood has to pump hard. It has to work as a turbo. So what happens is this, at that point, this is what we call the breaking point. Anything over 140. OK, and over 90.
[00:03:27] Systolic, diastolic. Now you’re running into problems that that engine has so much pressure to make up for the resistance.
[00:03:38] Yeah, OK. Yes. The overweight, the diabetic factor, the inflammatory factor. You’re talking about triglycerides over 150. You’re talking about type 2 diabetes. OK, again, type 2 diabetes, basically, you know, you’re not born with it. It’s something that you create. You create that diabetes. Where that insulin is out of balance. And now you’re talking about, again, a very large waistline, abdominal obesity. So a lot of times if you look at people, Alex, they look great.
[00:04:16] From the chest up. Yeah. And that mid-abdomen. That torso is scary. Yeah. Yeah. It’s a showstopper, as they say. OK. So this is where the high blood pressure comes in because again, that abdominal aorta, that pressure on it puts so much pressure that it goes above 140 and sometimes it goes over 180, which is like critical, critical. And again, with these characteristics, again, it creates abdominal cholesterol. It creates blood glucose over. We mentioned that over a plus 100 and again, high blood pressure connected with what stroke? So you have triglycerides.
[00:05:15] Clogging up. Triglycerides and yes, this is huge when the arteries clog up.
[00:05:21] Yeah. We have an issue with ultimately all the roads metabolically lead to the liver. Right. So one of the things that we’ve noticed is that when we assess the liver, sometimes they look pretty good and the symptoms may be highly elevated. There’s a huge range of liver enzymes. But what we’re noticing is that if we start having a blood sugar that is elevated. If we start having enzymes, if we start having disorders like what we call nonalcohol, this was a new disease that actually just came aboard. We always knew about alcoholic liver disease or cirrhosis. Now we have nonalcoholic liver cirrhosis and liver disease. Now, how did that happen? Because our sugar was too high and the triglycerides add these fats into the fat level of the liver and start actually destroying the liver. So we started having this disorder and it’s a huge issue, as you indicated, when we start having HDL levels and that which we measure, we start noticing greater levels in 40, lower levels than 40 for men and 50 for women. We start noticing little trends. We also start noticing abdominal obesity, high blood pressure. There are other areas like ovarian cysts.
[00:06:35] Mario, you’ve noticed that there are other areas that are indicated that are collaborative or even equal or what we use to determine metabolic syndrome.
[00:06:44] What are the ones you have noticed? The two major ones where we’re looking at the studies that you see. I’m going to pull this up for you. So we can kind of get a grasp on that. We’re looking at two just very simple.
[00:07:04] Let’s make things simple for the listeners and viewers. Number one.
[00:07:11] Right away. You’re talking about.
[00:07:15] Abdominal obesity. OK, that’s number one. Number two, insulin resistance. So what happens is that your whole sugar balance within your system is not tolerating, the insulin is not effective in your body. So this is where people, Alex, are always hungry and are always eating. And so what I call it. It’s almost like you’re overeating and you’re starving at the same time. Yeah. Because that sugar is in your blood vessels. Okay. And it’s not being taken into the cells. So the cells are starving. But your whole body is overeating. Does that make sense?
[00:08:01] Mario, you know, in your practice. Like, how many people? What percentage of people? Do you notice that even have metabolic syndrome?
[00:08:10] Just a scan and again, the literature says 23 percent of the population now, I would say and in our past community here, I would say at least double that to 40 to 45. And it’s really, really a point. This is why we’re here today. You know, we’re here to educate, inspire and most of all, give people simple understanding and solutions on what to do. And one thing that I can tell you with a lot of my patients, number one, increase your fiber intake. Like, eat more vegetables. You know, I tell them that. So what do you eat? Yeah. OK. You know, so, I mean, you come in and people want to get on treadmills and they want to climb mountains. They want to do burpees. It’s like, yeah, you know what? You’re 100 pounds overweight. Those burpees are going to kill you. Okay. You’re going to wake up tomorrow morning. You can’t get out of bed. So the major factor to really start to address this is not getting in and starting a workout in gyms. First, we’ve gotta handle and we need to really educate the public on, the food intake is the solution. That is the primary solution. Medicine of the body. The food is primary medicine. And this is what we’re talking about, becoming more vegetarian, increasing fiber, reducing alcohol intake. I know I’m hurting some folks right now. Yeah. Yeah. And the point is, you know, again, reducing. It doesn’t say, you know, if you want to have a beer or something, that’s great. But again, let’s be mindful. This metabolic syndrome is a beast. OK.
[00:10:07] It’s a beast that’s affecting our parents, our grandparents. And now, Alex, I can see these patterns in elementary school kids, OK? And what they’re eating, they’re eating a lot of sugars, right? They’re eating a lot of fast foods, processed foods. This is one thing like right now I’m looking at you and you’re drinking this green. Green, you know?
[00:10:32] Yeah. There you go. Yeah.
[00:10:35] It’s like a jolly green giant drink. OK. That is a live food. It’s uncooked. It’s raw. The vitamins are there. The nutrition is there. It’s not denatured. OK. Anytime we cook food, you kill it. Right. Anytime you bottle it up and you preserve it for a month and two months.
[00:11:02] I can tell you right now, you are not eating live. So the rule that I share with my patients and when I do seminars all over the nation and people invite me over, I say, look, if you want to be alive, why are you eating dead? Simply, why are you eating dead? Right. And like right now, you know, let’s make it simple. Number one, increase hydration. Right. OK. Have a gallon of water or more is a must. Excellent. That’s number one. Number two, increase live foods. Live foods are what? Vegetables. Fruits, right? Juice them. Eat them.
[00:11:38] I mean, from what I understand and what I do is that everything leads to, any sort of nutritional component, whether it’s a treatment for diabetes or a treatment for, let’s say, rheumatoid issues when there is a nutritional component, many times it’s hard getting the foods that you need. So the world has turned on smoothies. Smoothies can date different formats. And these smoothies, as we work them, ultimately have the solution. Now, what kind of smoothies? What’s the best type of approach with them? Well, we have that stuff in terms of awareness in our offices and gladly we’ll share that stuff whenever it’s necessary. However, the reason is that those inner parts of the cells, those living enzymes, those DNA molecules, those proteins, those…
[00:12:37] Actually, probiotics, even in the fiber, prebiotics, because when you do these smoothies or even do juicing that fiber you eat, that fiber that sometimes is lost while you do it juicy, juicing. This is important for the bacteria. So that helps even with disorders, such as leaky gut or intestinal dysbiosis, because they all come together. Someone who has metabolic syndrome most likely has leaky gut and vise versa. And not every single time, we can assess that, but what we want to do is we want to assess a person completely in terms of the drugs. You pretty much know that your doctor is trying to get you away from diabetes because you’re in that losing control place when you’re taking metformin.
[00:13:20] Metformin is very powerful, it’s a special medication that ultimately guides to restores the blood sugar back to where or it makes it more effective or makes you less insulin tolerant and more sensitive. So there’s a lot of things that we’re looking at that are useful for us. But one of the things is, well, what am I going to do with this?
[00:13:41] How am I going to get better? Well, diet? Diet has everything to start with. You start with your diet. You start with being vegetarian. You start with Mediterranean style foods. What kind of techniques do you use in terms of diets? Because I can go off and explain those things. But I want you to kind of get. Simple.
[00:13:56] You know, simplicity is golden. Complexity is chaos, Alex. The more complex we make it for ourselves, the more likely it is that we’re going to quit. OK. You can’t sustain complex things. We need to make them simple. So number one, as much as possible, eat raw, eat live foods. That’s number one. Number two. Number two, stop eating processed things like you’re talking about like high corn sirup, like simple, stop drinking Cokes and all of these fruity drinks and everything else, you know. And it tells you right there on the bottle, right there on a can. It’s 10 percent fruit. Do you know what 10 percent means? It’s not 100 percent. There’s a missing zero, baby. OK. It’s missing. So you know what? After a while, you’re gonna be missing off the planet. OK. You’re going to be extinct. Yeah. So, yeah. We need to get real, you know, this is real stuff. Like I can tell you, you know, I visit, I do home visits sometimes because I give back to the community and I go to families and I sit there and, you know, people are losing legs. People are losing limbs. They have wound care, you know. They have, you know, they have issues in terms of that. And that’s painful, not only for the person but for the whole family. So you know what? I take this very seriously. We take this very seriously at the show. You know, we want to make it live. We want to make it interesting. But I want to tell you right now, I’m not playing games. This is not game time. This is showtime. And so in terms of that, get off the Cokes, get off the fruity drinks, get off the candy bars. OK. You know, I think that commercial with Snickers, they need to change that. You know, instead of the Snickers satisfy, how about the Snickers gonna kick your ass? How about that one? Yeah, yeah. I said it. Thank you. I think we are physicians so we can say that word. Right. So that’s what’s happening. And then again, the lifestyle. If you’re drinking, the two things I can tell you right now. Yeah. Two things that are going to kick you in your gut, no pun intended. Metabolic boys and girls. OK. Number one is going to be smoking. And number two is going to be alcohol. You do those two.
[00:16:16] OK. And here it is. I’m going to tell you what it’s, you know, can you find that on the slide. OK. It’s gonna be the statistic. Oh, here it is. There. Bam, right there. Pull that up. Yeah, ok. Bam. OK. So if you’re looking at that, I’m going to tell you this is scary. Lifestyle factors, the two factors right there. Can you enlarge that? So we can see that. We can do that. OK. Excellent. I appreciate that. That’s all big. There it is. So now watch this. Do you see this? OK. Here it is. The number one life factor right here. Number one, is that? Yes. The one right here? OK. Now watch. I want to circle this. OK, where is it? It’s right there. Tools. I’ll get it for you. OK. Go ahead. Circle that, smoking and heavy drinking. Okay. I just want everyone to kind of take note, smoking and heavy drinking. That is one of the most destructive things that you can do right now. And guess what? Most people do them together, don’t they? Yes, sir. That’s it. So now watch, the ratio that metabolic syndrome affecting the man. OK. This is, again, something new. Mm-hmm. It’s affecting the man less than the women. Do you see that, guys? Yes, I do. Look at that. The women is 4.45. The women are affected most out of everyone. Out of everyone. And the men are at1.85. Now, the lesser evil is heavy drinking and poor diet, and the less one is smoking and physical activity. But if you really look at it, that’s what’s scary, smoking and heavy drinking. And this has really come down to a shift.
[00:18:04] You know, you used to be that men smoked and men drink. Now it’s changed, Alex. This is scary because it’s affecting, you know, momma is the boss. And to me, you know, mom is the doctor in the house. OK. And no, I don’t want to take responsibility for the man because you know what? We need to be the head, not the tail, but at the same time, who is going to take care of the kids?
[00:18:28] It’s got to be mama. You know, most of the time. Who’s going to take the kids to the doctor? Who’s gonna be wrong? So we need moms healthy. We need moms healthy. OK. We can, you know, because there’s a saying that says this, Alex. When mom ain’t happy. Dad ain’t happy.
[00:18:44] Nobody’s happy. No, thank you. Here, even the dog ain’t happy, Alex. He’s leaving now. He’s gone. He’s gone.
[00:18:52] I happen to know a lot of, I think after about 40, I think that in general there’s a tendency for the love of wine and it gets a little crazy for a wine to three a night. Yeah. This leads to metabolic syndrome. So we need to. Moderation is key. Right. Yeah. So we’re going to you know, if you’re if one of the biggest treatments is cardio, well why give yourself those extra calories and doing their process?
[00:19:19] Now one glass is fine. I understand that. But we don’t have to go crazy with the wine in the evening because it’s a more relaxing thing. You know, there’s always women’s night out, right? You know, I mean, it’s shot here, but women’s night out. You know, and when there’s women’s night out. And for many women, it’s a little bit of vino. So we need to kind of cater to those things, shall I say.
[00:19:39] And it’s woman’s night out. But it’s not all night, baby, you know?
[00:19:42] Yeah, OK. I mean, you know, there is.
[00:19:45] Exactly. I mean, you know, let’s have a glass but not the bottle, baby. Come on. Well, you know. Right. If you have 4 people a bottle is OK. OK. I said wine. Okay. I’m sorry. I lost. I lost the meaning of that. Go ahead Alex. Yeah.
[00:19:57] So the bottom line is, that we’re here to bring in awareness of this disorder, which is metabolic syndrome.
[00:20:05] Honestly, I’ve been going to school for a long time. And this is a new revelation of the last decade.
[00:20:12] The gastroenterologist is really focusing on. They’re the first to see it, the dimensions of. And here’s the thing. Crazy metabolic syndrome leads to nonalcoholic, fatty liver disease. And you were seeing this in rampant levels, literally. Gallstones. Exactly. Because the liver, the cholesterol issues, all these dynamic changes are affecting even our children. We’re having kids with nonalcoholic fatty liver disease. Why? Too much sugar? Too much sugar? We have to control the sugars. And there are things that we can do in our diets. Plan on bringing all those concepts.
[00:20:46] But we want to bring awareness as to what happens. Lack of sleep, cortisol raising, you know, all this kind of stuff alters the blood sugar in our system. So it’s very important to do the best we can. Exercising is awesome for this stuff in terms of cardiogenic exercises, cardiometabolic dynamics, though. That’s where we want to still focus on. We have to do a little bit of cardio. We got to, you know, eat more of vegetables, greens, juicing, those kinds of things sleep better. It’s important to sleep.
[00:21:15] Alex. Oh, OK. I want to jump in because I know we’re jumping here, you know, and people and I like the fact what you mentioned earlier, you know, we want to, you know, get some wine and some things to relax. Why don’t we do this? I encourage people to meditate. OK. And to try to create some stress management strategies. OK. Right.
[00:21:38] Like a nice, you know, nice warm cup of tea an hour before we go to sleep. You know, some chamomile tea and I mean, you know, chamomile, you know, and.
[00:21:50] Yeah. Good stuff. Yerba buena. You know, stuff like that. You heard about it? Yeah. Yeah. So.
[00:21:56] So you know, all of these things. Meditation because why? Stress levels, as you mentioned, I’m on one accord with that stress level increase cortisol, which contracts, arterial function and then decreases dopamine.
[00:22:13] OK, and oxytocin, which is like the love thing going on. OK. And so now all of this creates sleep apnea. And how many people do you know, Alex, that suffers from sleep apnea and instead of dealing with the causation? OK. So this is why chiropractic is such a beautiful thing.
[00:22:35] You know, for 25 plus years, a quarter of a century plus, you know, between both of us, we’re like, gosh, 60 plus years. Correct? Yes, 60 plus years. All right. Chiropractic and chiropractors have such a beautiful story and beautiful platform because we’re all about natural healing and helping our community and our country at very, very cost-effective.
[00:23:09] You know, we do one of the most cost-effective ways of not only treating health but preventing health. And we are ambassadors of health. I mean. And so this is where when we’re talking about, again, sleep apnea, meditation, I see so many people in my practice, they’re taking a pill to go to sleep.
[00:23:32] Every night.
[00:23:34] They suffer from depression. They suffer from anxiety. Okay. And then I look and I go, you know what? Let’s talk about your lifestyle. What are you doing? What are you doing every day to put your body in a high, intense, inflammatory system in high, intense stress? Yeah, you’re redlining. I always tell people. Right now you’re redlining it’s just like a car. You cannot maintain it. You better change gears, otherwise, you’re going to blow the engine. And this is what I see, sleep apnea. Sleep is again, all the way from athletics to life function. That is where the neuroreceptors neuroplasticity, Alex. Okay. That’s where we heal. We recover. We reset for the next fight, which is early in the morning. And if we don’t do that, we go to the next day with that fog. Yeah. You know that mental fog. Alex. Hey, you know, and this is where people say, you know, I can’t focus. I’m forgetting, you know, and I don’t know what’s happening, you know, and I’m going, you know why you’re not sleeping?
[00:24:47] Exactly. You know, we’ve done the studies and the studies specifically about sleep. If you’re a person that you need seven hours of sleep and you miss one hour, just one hour of sleep.
[00:24:58] The mind is very just speaking about just the brain fog that happens with this metabolic syndrome because it really starts disrupting everything. One hour of sleep actually diminishes your ability to be creative. Creativity. Yes, the brain is altered. And you may think that you know, 25 percent by one hour, it makes a difference. But literally two hours of sleep, you lose 50 percent of your creativity if you lose four hours of sleep. Well, no one of those when you just push it like four hours. Your creativity is so low that even just trying to figure out how to find where your keys are or creativity, how to solve problems, how to deal with issues, you go to work and someone’s got some drama. It stresses you out and metabolically and then what would a lot of people do? You go to work. You jack yourself up with some coffee and then you grab what? Many people grab the donut instantly, send in the blood sugar into chaos. This constant repetitive issue of habits leads to the disorder of metabolic syndrome.
[00:26:02] The body, the body. Alex cannot sustain us. You know, you can’t. As I mentioned before, you can’t drive that, that your car in first gear going 80 miles an hour. You cannot. And so it’s going to rip at the seams. The gasket is going to come off. The oil is going to spray out. OK. And this is what’s happening. If you really look at the body, we look at the HDL cholesterol, it’s lower than 40. They should be the highest, high density.
[00:26:33] High density should be the highest. Cholesterol, good cholesterol stuff.
[00:26:37] You know, you want, LDL should be low and the HDL should be high. You’re looking at insulin. You’re looking at strokes. You’re looking at again, you know, triglycerides over 150, you know, sleep apnea again, more than ever. Now I’m hearing about it. Polycystic ovaries. I just hear this, you know. You know, I have another surgery. You know, I have cysts.
[00:27:04] I have this with all of these things. If we really look at it, we are seeing that we are pushing ourselves over the cliff. Yeah, OK. And then I’m going to throw this in there with the onset of overuse of cell phones by adults and by youth. Now it is disrupting the cognitive pattern, the focus pattern. And it’s throwing people where they don’t go to sleep because they have a cell phone in front of them at 11 o’clock at night in bed. You know, and then you’re talking about like one hour less than two hours. Well, let me tell you, the worst thing you could do, Alex, is have your cell phone next to your head with the radiation, with the sounds every time that phone beeps. Tweaks a bell, sounds for an email or your Facebook. Guess what your subconscious in your brain does? It wakes up. It wakes up and bam, it blows up.
[00:28:11] That REM sleep you only have. What is it, Alex? What, ten, 10 minutes, less than 15 minutes, of REM sleep? Oh, there’s a lot of. What is it?
[00:28:20] What happens is the body goes into a bunch of different stages as it does. What we’ve learned over the years is that we used to believe that REM was something intermittent. It goes into these deep, deep levels throughout the night.
[00:28:31] Your body temperature, what your blood sugars at, what’s your mental state, what’s your tired levels is, what your electrolyte balance is. This matters. So sleeping is very important. So in the restoration of your body through to get it back in order. Sleep is one of the greatest ways to be able to restore the body. So it’s important to try to you know, if you go to bed at 10, push yourself to start working the process, to go to bed probably about an hour earlier or start working. And the TV is going to be there. They’re going to keep on going 24/7. But, you know, we’re the ones dealing with the issue later on in the next day where we are brain doesn’t work well.
[00:29:10] Our bodies are needing carbohydrates, our foods are. And don’t eat, you know, and put yourself on one of the things that I encourage. Again, my children and, you know, Karen and the kids. Is this. Put your cell phone in the kitchen and get a real alarm clock. I don’t think they make them anymore, do they? They don’t make alarm clocks anymore. Now, forget it. What’s an alarm clock? It’s kind of like a pager, Alex. You know what? We’re gonna get to the point where we’re going to say, hey, do you have an alarm clock? And I go, now I’ve got a pager. All right. Okay. It’s ridiculous. We need to have a phone in the kitchen. You remember back in the days with that long cord. About like 80 yards. You used to take it in your room for those private sessions, you know, with your girlfriend and all of that.
[00:29:58] Well, let me tell you, that cell phone needs to be in the kitchen. It needs to be turned off. Okay. And then you need to have an alarm clock next to your bed. You need to honor sleep. You cannot eat foods before you go to sleep. Go to sleep hungry.
[00:30:16] One of the things that I’m reading more and more in terms of research and in terms of data. Go to sleep, hunger, you’re not going to die. All right. Calm down. Well, I know what you’re talking about. Yes. Intermittent fasting. Absolutely. OK. Yeah. Yeah. You can’t. I mean, at the end, you know, let’s say 8:00. That’s enough. Put the burger down.
[00:30:35] Yeah. Look at that. You know.
[00:30:37] You know, 300 years ago, we would be we many of us were nomadic. You know, people live culturally on the sides of the earth. And the terrain was different. We got the food during the daytime. Nighttime was a time when you kind of just, you know, settled in. All right. If you did, have you had some grains, some nuts, and it was different. So just by using the sunlight in order to kind of like you, as soon as the sun comes up, you can eat and shut off at night if you get really good and you start using what we’re learning, that is a great method, which is intermittent fasting. The body has the ability to rectify itself. This is an amazing, metabolic syndrome or not. The body even activity stream has the ability to rectify itself.
[00:31:23] So if you allow the body to cleanse itself throughout the day, let’s say you do an eating cycle of only eight hours on a window, so to speak. Well, you got a good, you know, 16 hours of what period of where your body breaks things down. Well, the metabolic processes of the breakdown of usually the mitochondria, the mitochondria, as it starts working, the process needs to rest. Our bodies need to rest. I mean, if I told you to cut the grass, Mario, you know, cut the grass. And as soon as you walked in the house and you were done and I gave you some lemonade and you were just kinda, that was rough. And I said, go back and cut it again. Right. And then also you go back out and you’re like, wow, this is crazy. When what kind of guy? And then right when you’re tired and you’re shaking because you have no energy and you’re about to go to bed. All right. And you go get up because you’re gonna go cut the grass again and you never let the body rest. Eventually, your body breaks down. That’s what happens with mitochondria. If you’re constantly eating, you’re constantly going through a process, burn out, burning, burning and burning. It needs time to settle and relax, to recover. And that’s what metabolic syndrome recovery process is. Do we try to calm the body through sleep, through lifestyle changes, through blood sugar issue changes? And little by little, we’ll start getting back control of your life because otherwise you have increased steroid or what we call a cortisol steroid production, which then makes the body store fat. Right. And then that’s where you get the belly issues because one of the first indicators of metabolic syndrome is a big belly. Right. For a woman, it’s over a certain amount, over 35 inches. And for men, anywhere over 40, that’s a lot of people over 40 inches. You pretty much have a kickoff towards metabolic syndrome.
[00:33:00] And how many times have you heard in your practice, Alex? You know, people come in and go, you know, I just need to lose weight. I just need to lose weight. And, you know, I look at him and I go, you know, forget about the weight.
[00:33:14] What are you doing?
[00:33:15] Are you sleeping well? All right. Right. Exactly. I mean, first. I mean, let’s forget about the weight. The body knows how to calibrate, Alex. It knows how to calibrate. It knows the zone, the sweet zone, the target zone. It knows genetically DNA, RNA. We have an innate intelligence. OK. We learned this in school and chiropractic school.
[00:33:38] The body has innate intelligence to know how to heal, to know how to recover, to know how to grow. And to know how to survive when we put undue stress to our system, to that fiber optics, to that circuit, we blow it and then it goes a wall. And that’s what happened. So I tell people, you know, no, the answer is not for you to go to a gym for 20 hours a day. No. Why don’t you start with letting go of high corn, syrup high fructose corn, syrup drinks?
[00:34:17] Why don’t you do that first? That’s what I tell them right away. Do that first. Number two, eat more fiber. What does fiber mean? Eat more legumes. Spinach, OK. Like broccoli. And instead of cooking them. And I know most people don’t want to eat raw. Why don’t you just steam them? OK. Just kind of steam them a little bit. OK. Don’t fire. Don’t kill them. Steam them. The other thing you want to do is, you know, get up on a Mediterranean diet, you know, fresh seafood, things like that, regular exercise, just get up in the morning. The first thing that I would share with people is just make things simple.
[00:34:53] Wake up when the rest of the animals wake up, OK? Wake up. And just go for a walk.
[00:35:02] At least, you know, let’s not get on a treadmill and break a record, OK? Let’s not do that because you’re gonna do that twice. You’re gonna get hurt and you’re gonna quit for the next three months to recover. So why don’t you just get up and walk? And then next time walk a little more and walk a little faster and next time walk a little faster and jog. So we need to create that patterning, rituals. We need healthy rituals, Alex, in our lives instead of rituals that are killing us slowly. OK. And so in terms of that, I look at eating more fresh vegetables, Mediterranean, sleep better and honor that sleep. Get up earlier, get up before 5 o’clock. OK. Get up. Get into a routine that way when you when you’re not sleeping late. OK. Your body’s tired. So now you can go to sleep at nine-thirty, at ten o’clock, OK. And then create a new pattern to where you let go of the cell phone. And I would say, you know how people do like fasting or cleansing. I recommend at least two cleanses a year, two cleanses a year. You know, stop eating meats.
[00:36:15] OK. Stop eating meats.
[00:36:17] And just go vegetarian. Okay. Let your body cleanse, I know you have wonderful programs at your clinic. Alex, in terms of detox, I believe in that twice a year. You gotta do it. It’s just like flushing your car. Alex Right.
[00:36:31] From detoxing. We start we really begin the process at the intestine. So from the things we start eating. So one of the best ways to do it is to go through a detox program and the detox programs help us cleanse it.
[00:36:45] You know what? If your garage is full of mud, if you’re you know, you just open the garage, you see it’s just a mess and your body’s metabolically a wreck similar to a garage as an analogy. Well, you can have a couple of things. The first thing we do is we open up the garage and we got to plunge that garage. We’ve got to get the hose. Or you can take the broom. And you know what? Many times you’ll realize that taking the hose is probably the best thing. So when we do detox programs, we begin by helping with the colon cleanse. And we basically clean out the colon and eventually also repurpose the direction where the probiotics go. We actually add probiotics in the diet. We add the fibers, the prebiotics, the post probiotics. And we start working from the intestine out. The body instantly starts recovering. And little things that we can do in the mornings on a regular basis such as increase water, take a lemon, for example. We start with water. We start with let’s say you squeeze four or five, three lemons, just three lemons, squeeze the juice, put it in a drink that you’re gonna spend all day. Take one whole lemon to squeeze it nice and just take it as a shot. Some people use apple cider vinegar. Exactly. So what we do is we start that well, that goes into the intestines, into the stomach. It is much it just basically showers the liver with a process that begins the process of cleansing as the body starts recovering. You start eating better. You let’s say you do a fast and you begin your process of eating at around 12:00 and you stop at six or maybe seven or maybe eight. And it gives you an eight-hour window, six to eight-hour window and that’s it. And then you start recovering in that process. So early on, we start doing the changes with the beautiful thing about the body if you give it time, it’s all recoverable to do it. And if you teach the body, you don’t have to be taken metformin the rest of your life. Now, if your doctor thinks it’s appropriate, well, then, you know, you speak to your doctor. And here’s the other thing is, is that when you’re dealing with metabolic issues and you’ve got someone that’s giving you metformin and as it or other diabetes or different types of medications that help with diabetes, it is appropriate to have it under a doctor’s control. Now, the constant taking medications without, having lifestyle changes or not having dietary alterations is inappropriate. It’s malpractice, I believe, in my opinion, after being here so long, you just don’t give people pills and not change their diet. You got to have a doctor who’s willing to sit down and help you out with lifestyle changes, diet or someone that has some staff to support you or a dietician or a registered dietetic dietitian. So that can help you or a nutritionist. Sorry. Or a health coach that can help you. We have to have people that can do that. So to help you with the process, because it’s not just saying, hey, take a pill and go off on your own, that confuses a lot of people. We have to have a change and we’re here to help you with that. So as we go through this process over the next couple of weeks, Mario, as we discuss each one of these particular topics, we’re going to bring it to you. We’re going to talk about ways that you can change your diet that simple. Just make it simple and it adapts. So I know you’ve got some points you want to mention again.
[00:39:50] Absolutely. You know, with metabolic syndrome, the two things that are markers are number one, your belly fat. OK, so let’s get going in at home. Yeah. Let’s wrap it up. You know, let’s button this thing up and make it simple. First of all, it’s belly fat. So. So if your belt, when you’re buying a belt and you’re looking at the little notches and the little holes and they’re over 40 inches, OK, 40, you’re in trouble. For male. Yeah, for male. And if you’re over 35 for a female, you’ve got issues.
[00:40:24] Metabolic syndrome. Absolutely. Absolutely. And the other point is this. If you have a hard time and you can’t even wear a belt, then I don’t need to explain any further. It’s time to come in and get a coach, get a health coach, get someone to make you accountable because time is ticking. Time is ticking and we need to be proactive instead of reactive. And at the same time, when you are always hungry, you’re overeating and you keep being hungry. You cannot lose weight. Now the things are coming to fruition. And on top of that, if you have blood pressure, over 140, over 90. So let’s say 145 over 100, let’s say 150 over 105. It’s time to get real. You can do this. Just go buy a blood pressure cuff, OK? Quit playing games. Get-go. Go to Walgreens. Get a blood pressure cuff and check it tonight. And if it’s over. Check it at least. I always tell people, don’t do it once. Keep a record and do it for at least three, three to four days. OK. And take it in the morning. In the afternoon at night so you can see a cycle. Bring that over and find yourself a team. Find yourself a team that will that has a program for you to naturally empower you and give you the right tools to get back to your best life ever. It is up to you. It’s called self-responsibility. No pill is going to take the responsibility from you. And at the same time, you can enjoy your family more, enjoy your life, and be happier in the process.
[00:42:19] So I really appreciate it, Alex. And I want to thank our listeners and viewers and we look forward to continue the conversation of health and wealth. Because I want to tell you, the message for today is your health is your ultimate wealth. Thank you. And we’ll leave it there.
[00:42:38] All right, guys. Thank you.
[00:42:39] And we’ll come back tomorrow and we’ll add another health nugget, so to speak, for you guys. Two miners.
Podcast: BIA and Basal Metabolic Rate Explained
Dr. Alex Jimenez and Dr. Mario Ruja discuss basal metabolic rate, BMI, and BIA. Body mass and body fat can be measured in a variety of ways, however, several measurement tools may ultimately be inaccurate for many athletes. According to Dr. Alex Jimenez and Dr. Mario Ruja, calculating an individual’s body mass and body fat utilizing various tools is essential to determine overall health and wellness. BMI uses a person’s height divided by twice their weight. The results may be inaccurate for athletes because their body mass and body fat are different, in terms of weight, compared to the average person. Dr. Alex Jimenez and Dr. Mario Ruja demonstrate that BIA, or bioelectrical impedance analysis, and various other tools, such as the DEXA test, the Tanita scale, and the InBody, among others, can help more accurately determine an athlete’s body mass and body fat. Basal metabolic rate, BMI, and BIA are essential for parents that have young athletes as well as for the general population. Healthcare professionals that have these tools available can ultimately help provide individuals with the results they may need to maintain overall health and wellness.
Neural Zoomer Plus for Neurological Disease
Dr. Alex Jimenez utilizes a series of tests to help evaluate neurological diseases. The Neural ZoomerTM Plus is an array of neurological autoantibodies which offers specific antibody-to-antigen recognition. The Vibrant Neural ZoomerTM Plus is designed to assess an individual�s reactivity to 48 neurological antigens with connections to a variety of neurologically related diseases. The Vibrant Neural ZoomerTM Plus aims to reduce neurological conditions by empowering patients and physicians with a vital resource for early risk detection and an enhanced focus on personalized primary prevention.
Food Sensitivity for the IgG & IgA Immune Response
Dr. Alex Jimenez utilizes a series of tests to help evaluate health issues associated with a variety of food sensitivities and intolerances. The Food Sensitivity ZoomerTM is an array of 180 commonly consumed food antigens that offers very specific antibody-to-antigen recognition. This panel measures an individual�s IgG and IgA sensitivity to food antigens. Being able to test IgA antibodies provides additional information to foods that may be causing mucosal damage. Additionally, this test is ideal for patients who might be suffering from delayed reactions to certain foods. Utilizing an antibody-based food sensitivity test can help prioritize the necessary foods to eliminate and create a customized diet plan around the patient�s specific needs.
Gut Zoomer for Small Intestinal Bacterial Overgrowth (SIBO)
Dr. Alex Jimenez utilizes a series of tests to help evaluate gut health associated with small intestinal bacterial overgrowth (SIBO). The Vibrant Gut ZoomerTM offers a report that includes dietary recommendations and other natural supplementation like prebiotics, probiotics, and polyphenols. The gut microbiome is mainly found in the large intestine and it has more than 1000 species of bacteria that play a fundamental role in the human body, from shaping the immune system and affecting the metabolism of nutrients to strengthening the intestinal mucosal barrier (gut-barrier). It is essential to understand how the number of bacteria that symbiotically live in the human gastrointestinal (GI) tract influences gut health because imbalances in the gut microbiome may ultimately lead to gastrointestinal (GI) tract symptoms, skin conditions, autoimmune disorders, immune system imbalances, and multiple inflammatory disorders.
Formulas for Methylation Support
XYMOGEN�s Exclusive Professional Formulas are available through select licensed health care professionals. The internet sale and discounting of XYMOGEN formulas are strictly prohibited.
Proudly,�Dr. Alexander Jimenez makes XYMOGEN formulas available only to patients under our care.
Please call our office in order for us to assign a doctor consultation for immediate access.
If you are a patient of Injury Medical & Chiropractic�Clinic, you may inquire about XYMOGEN by calling 915-850-0900.
For your convenience and review of the XYMOGEN products please review the following link. *XYMOGEN-Catalog-Download
* All of the above XYMOGEN policies remain strictly in force.
Modern Integrated Medicine
The National University of Health Sciences is an institution that offers a variety of rewarding professions to attendees. Students can practice their passion for helping other people achieve overall health and wellness through the institution’s mission. The National University of Health Sciences prepares students to become leaders in the forefront of modern integrated medicine, including chiropractic care. Students have an opportunity to gain unparalleled experience at the National University of Health Sciences to help restore the natural integrity of the patient and define the future of modern integrated medicine.











 We usually talk of energy in general terms, as in �I don�t have a lot of energy today� or �You can feel the energy in the room.� But what really is energy? Where do we get the energy to move? How do we use it? How do we get more of it? Ultimately, what controls our movements? The three metabolic energy pathways are the�phosphagen system, glycolysis�and the�aerobic system.�How do they work, and what is their effect?
We usually talk of energy in general terms, as in �I don�t have a lot of energy today� or �You can feel the energy in the room.� But what really is energy? Where do we get the energy to move? How do we use it? How do we get more of it? Ultimately, what controls our movements? The three metabolic energy pathways are the�phosphagen system, glycolysis�and the�aerobic system.�How do they work, and what is their effect?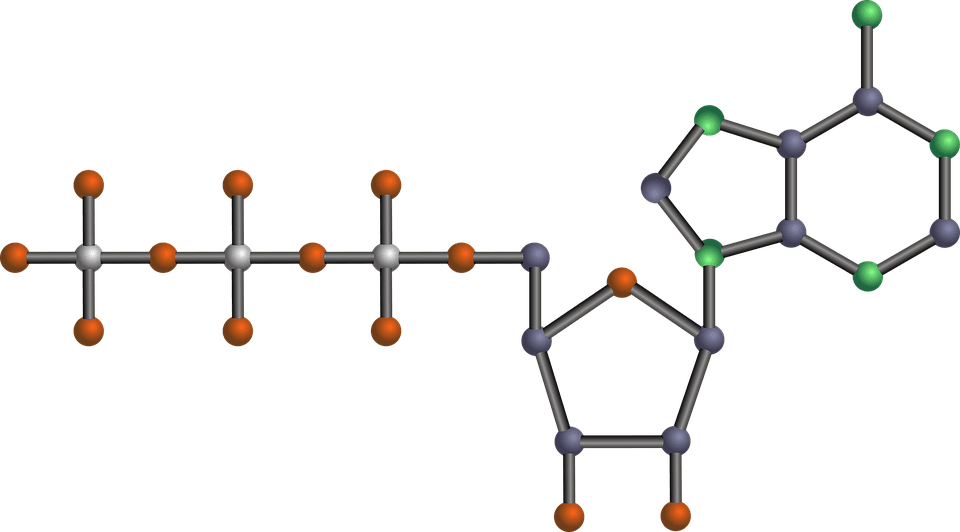 The energy for all physical activity comes from the conversion of high-energy phosphates (adenosine�triphosphate�ATP) to lower-energy phosphates (adenosine�diphosphate�ADP; adenosine�monophosphate�AMP; and inorganic phosphate, Pi). During this breakdown (hydrolysis) of ATP, which is a water-requiring process, a proton, energy and heat are produced: ATP + H2O ���ADP + Pi�+ H+�+ energy + heat. Since our muscles don�t store much ATP, we must constantly resynthesize it. The hydrolysis and resynthesis of ATP is thus a circular process�ATP is hydrolyzed into ADP and Pi, and then ADP and Pi�combine to resynthesize ATP. Alternatively, two ADP molecules can combine to produce ATP and AMP: ADP + ADP ���ATP + AMP.
The energy for all physical activity comes from the conversion of high-energy phosphates (adenosine�triphosphate�ATP) to lower-energy phosphates (adenosine�diphosphate�ADP; adenosine�monophosphate�AMP; and inorganic phosphate, Pi). During this breakdown (hydrolysis) of ATP, which is a water-requiring process, a proton, energy and heat are produced: ATP + H2O ���ADP + Pi�+ H+�+ energy + heat. Since our muscles don�t store much ATP, we must constantly resynthesize it. The hydrolysis and resynthesis of ATP is thus a circular process�ATP is hydrolyzed into ADP and Pi, and then ADP and Pi�combine to resynthesize ATP. Alternatively, two ADP molecules can combine to produce ATP and AMP: ADP + ADP ���ATP + AMP. During short-term, intense activities, a large amount of power needs to be produced by the muscles, creating a high demand for ATP. The phosphagen system (also called the ATP-CP system) is the quickest way to resynthesize ATP (Robergs & Roberts 1997). Creatine phosphate (CP), which is stored in skeletal muscles, donates a phosphate to ADP to produce ATP: ADP + CP ���ATP + C. No carbohydrate or fat is used in this process; the regeneration of ATP comes solely from stored CP. Since this process does not need oxygen to resynthesize ATP, it is anaerobic, or oxygen-independent. As the fastest way to resynthesize ATP, the phosphagen system is the predominant energy system used for all-out exercise lasting up to about 10 seconds. However, since there is a limited amount of stored CP and ATP in skeletal muscles, fatigue occurs rapidly.
During short-term, intense activities, a large amount of power needs to be produced by the muscles, creating a high demand for ATP. The phosphagen system (also called the ATP-CP system) is the quickest way to resynthesize ATP (Robergs & Roberts 1997). Creatine phosphate (CP), which is stored in skeletal muscles, donates a phosphate to ADP to produce ATP: ADP + CP ���ATP + C. No carbohydrate or fat is used in this process; the regeneration of ATP comes solely from stored CP. Since this process does not need oxygen to resynthesize ATP, it is anaerobic, or oxygen-independent. As the fastest way to resynthesize ATP, the phosphagen system is the predominant energy system used for all-out exercise lasting up to about 10 seconds. However, since there is a limited amount of stored CP and ATP in skeletal muscles, fatigue occurs rapidly.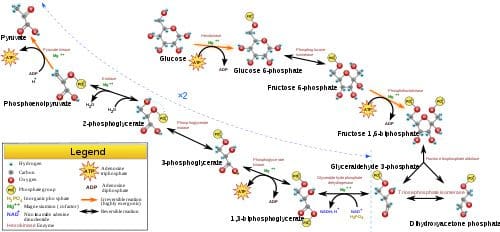 Glycolysis is the predominant energy system used for all-out
Glycolysis is the predominant energy system used for all-out  Since humans evolved for aerobic activities (Hochachka, Gunga & Kirsch 1998; Hochachka & Monge 2000), it�s not surprising that the aerobic system, which is dependent on oxygen, is the most complex of the three energy systems. The metabolic reactions that take place in the presence of oxygen are responsible for most of the cellular energy produced by the body. However, aerobic metabolism is the slowest way to resynthesize ATP. Oxygen, as the patriarch of metabolism, knows that it is worth the wait, as it controls the fate of endurance and is the sustenance of life. �I�m oxygen,� it says to the muscle, with more than a hint of superiority. �I can give you a lot of ATP, but you will have to wait for it.�
Since humans evolved for aerobic activities (Hochachka, Gunga & Kirsch 1998; Hochachka & Monge 2000), it�s not surprising that the aerobic system, which is dependent on oxygen, is the most complex of the three energy systems. The metabolic reactions that take place in the presence of oxygen are responsible for most of the cellular energy produced by the body. However, aerobic metabolism is the slowest way to resynthesize ATP. Oxygen, as the patriarch of metabolism, knows that it is worth the wait, as it controls the fate of endurance and is the sustenance of life. �I�m oxygen,� it says to the muscle, with more than a hint of superiority. �I can give you a lot of ATP, but you will have to wait for it.�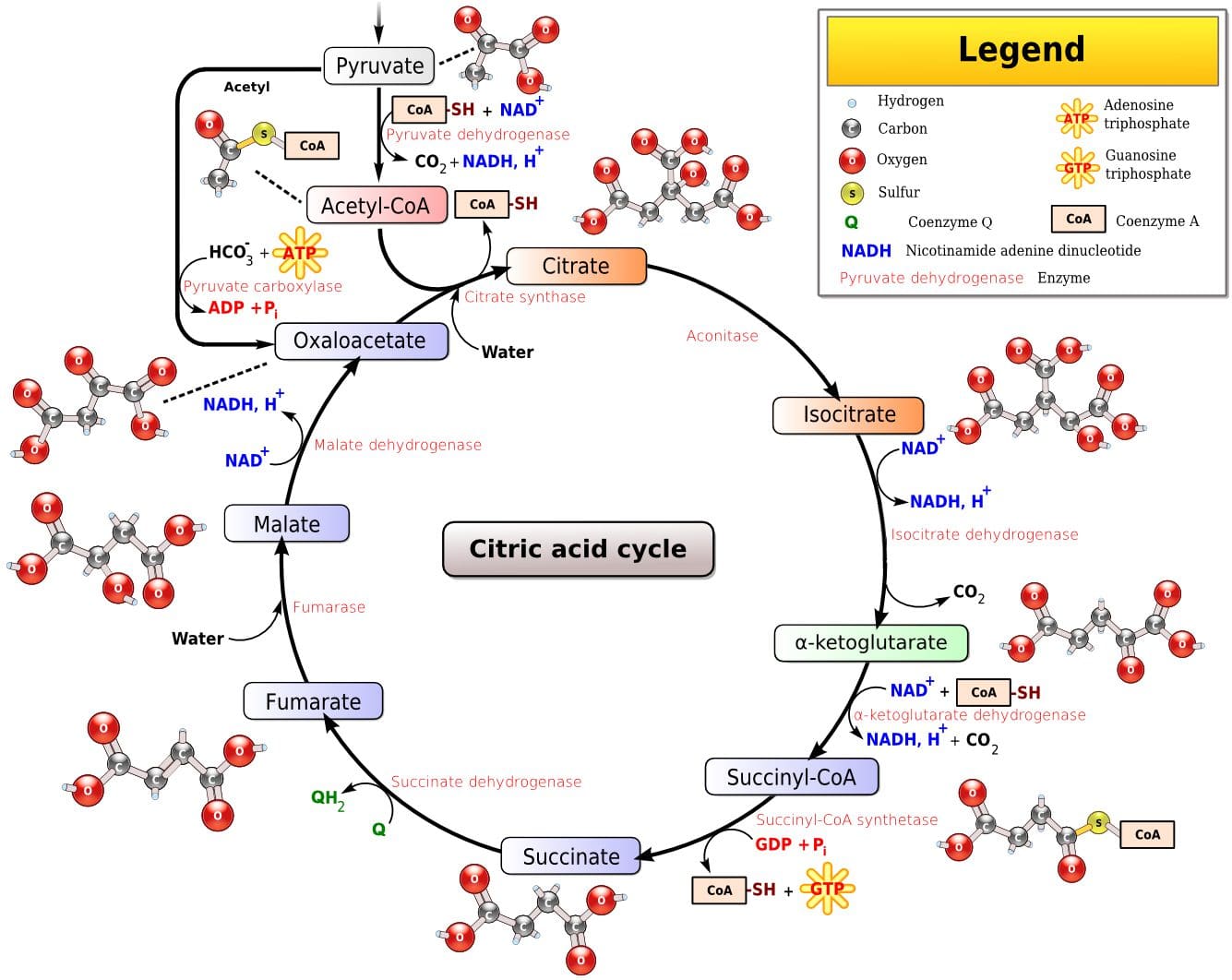 Fat, which is stored as triglyceride in adipose tissue underneath the skin and within skeletal muscles (called�intramuscular triglyceride), is the other major fuel for the aerobic system, and is the largest store of energy in the body. When using fat, triglycerides are first broken down into free fatty acids and glycerol (a process called�lipolysis). The free fatty acids, which are composed of a long chain of carbon atoms, are transported to the muscle mitochondria, where the carbon atoms are used to produce acetyl-CoA (a process called�beta-oxidation).
Fat, which is stored as triglyceride in adipose tissue underneath the skin and within skeletal muscles (called�intramuscular triglyceride), is the other major fuel for the aerobic system, and is the largest store of energy in the body. When using fat, triglycerides are first broken down into free fatty acids and glycerol (a process called�lipolysis). The free fatty acids, which are composed of a long chain of carbon atoms, are transported to the muscle mitochondria, where the carbon atoms are used to produce acetyl-CoA (a process called�beta-oxidation).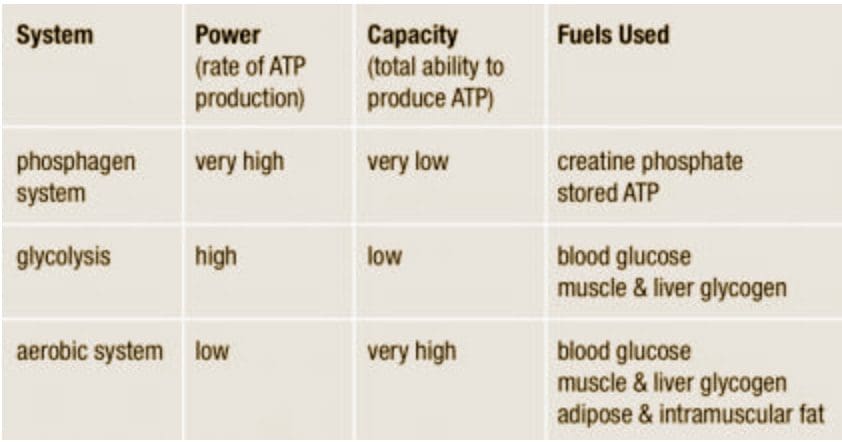


 It was during the period between 1910 and 1920 when it was suggested for the first time that a cluster of associated metabolic disturbances tended to coexist together [1]. Since then, different health organisms have suggested diverse definitions for metabolic syndrome (MetS) but there has not yet been a well-established consensus. The most common definitions are summarized in Table 1. What is clear for all of these is that the MetS is a clinical entity of substantial heterogeneity, commonly represented by the combination of obesity (especially abdominal obesity), hyperglycemia, dyslipidemia and/or hypertension [2�6].
It was during the period between 1910 and 1920 when it was suggested for the first time that a cluster of associated metabolic disturbances tended to coexist together [1]. Since then, different health organisms have suggested diverse definitions for metabolic syndrome (MetS) but there has not yet been a well-established consensus. The most common definitions are summarized in Table 1. What is clear for all of these is that the MetS is a clinical entity of substantial heterogeneity, commonly represented by the combination of obesity (especially abdominal obesity), hyperglycemia, dyslipidemia and/or hypertension [2�6].
 Several dietary strategies and their potential positive effects on the prevention and treatment of the different metabolic complications associated to the MetS, are described below and summarized in Table 2.
Several dietary strategies and their potential positive effects on the prevention and treatment of the different metabolic complications associated to the MetS, are described below and summarized in Table 2.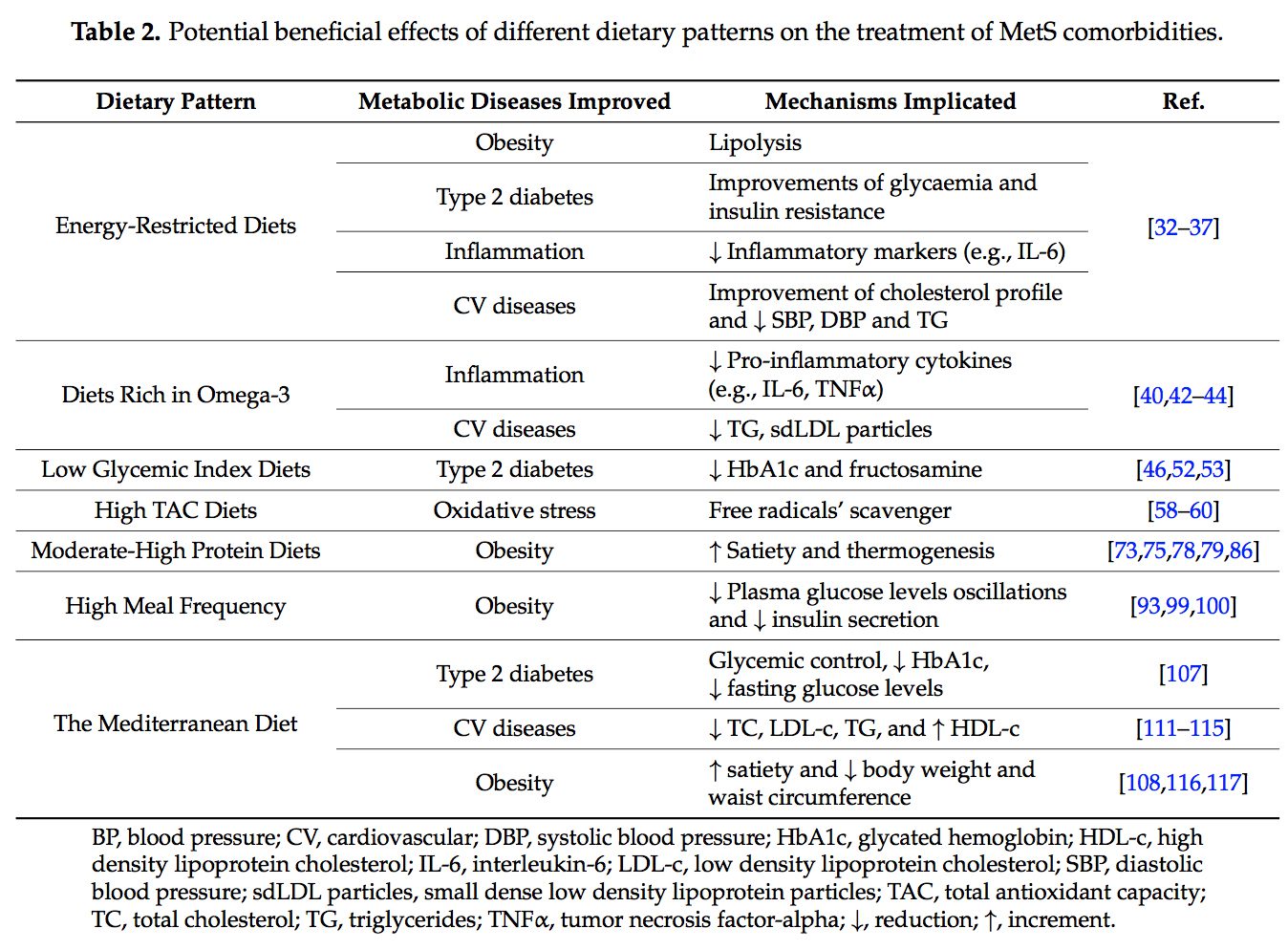 2.1. Energy-Restricted Diet Strategies
2.1. Energy-Restricted Diet Strategies
 The very long-chain eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) are essential omega-3 polyunsaturated fatty acids (n-3 PUFAs) for human physiology. Their main dietary sources are fish and algal oils and fatty fish, but they can also be synthesized by humans from ?-linolenic acid [40].
The very long-chain eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) are essential omega-3 polyunsaturated fatty acids (n-3 PUFAs) for human physiology. Their main dietary sources are fish and algal oils and fatty fish, but they can also be synthesized by humans from ?-linolenic acid [40]. Over the last ten years, the concern about the quality of the carbohydrates (CHO) consumed has risen [46]. In this context, the glycemic index (GI) is used as a CHO quality measure. It consists in a ranking on a scale from 0 to 100 that classifies carbohydrate-containing foods according to the postprandial glucose response [47]. The higher the index, the more promptly the postprandial serum glucose rises and the more rapid the insulin response. A quick insulin response leads to rapid hypoglycemia, which is suggested to be associated with an increment of the feeling of hunger and to a subsequent higher caloric intake [47]. The glycemic load (GL) is equal to the GI multiplied by the number of grams of CHO in a serving [48].
Over the last ten years, the concern about the quality of the carbohydrates (CHO) consumed has risen [46]. In this context, the glycemic index (GI) is used as a CHO quality measure. It consists in a ranking on a scale from 0 to 100 that classifies carbohydrate-containing foods according to the postprandial glucose response [47]. The higher the index, the more promptly the postprandial serum glucose rises and the more rapid the insulin response. A quick insulin response leads to rapid hypoglycemia, which is suggested to be associated with an increment of the feeling of hunger and to a subsequent higher caloric intake [47]. The glycemic load (GL) is equal to the GI multiplied by the number of grams of CHO in a serving [48]. Dietary total antioxidant capacity (TAC) is an indicator of diet quality defined as the sum of antioxidant activities of the pool of antioxidants present in a food [55]. These antioxidants have the capacity to act as scavengers of free radicals and other reactive species produced in the organisms [56]. Taking into account that oxidative stress is one of the remarkable unfortunate physiological states of MetS, dietary antioxidants are of main interest in the prevention and treatment of this multifactorial disorder [57]. Accordingly, it is well-accepted that diets with a high content of spices, herbs, fruits, vegetables, nuts and chocolate, are associated with a decreased risk of oxidative stress-related diseases development [58�60]. Moreover, several studies have analyzed the effects of dietary TAC in individuals suffering from MetS or related diseases [61,62]. In the Tehran Lipid and Glucose Study it was demonstrated that a high TAC has beneficial effects on metabolic disorders and especially prevents weight and abdominal fat gain [61]. In the same line, research conducted in our institutions also evidenced that beneficial effects on body weight, oxidative stress biomarkers and other MetS features were positively related with higher TAC consumption in patients suffering from MetS [63�65].
Dietary total antioxidant capacity (TAC) is an indicator of diet quality defined as the sum of antioxidant activities of the pool of antioxidants present in a food [55]. These antioxidants have the capacity to act as scavengers of free radicals and other reactive species produced in the organisms [56]. Taking into account that oxidative stress is one of the remarkable unfortunate physiological states of MetS, dietary antioxidants are of main interest in the prevention and treatment of this multifactorial disorder [57]. Accordingly, it is well-accepted that diets with a high content of spices, herbs, fruits, vegetables, nuts and chocolate, are associated with a decreased risk of oxidative stress-related diseases development [58�60]. Moreover, several studies have analyzed the effects of dietary TAC in individuals suffering from MetS or related diseases [61,62]. In the Tehran Lipid and Glucose Study it was demonstrated that a high TAC has beneficial effects on metabolic disorders and especially prevents weight and abdominal fat gain [61]. In the same line, research conducted in our institutions also evidenced that beneficial effects on body weight, oxidative stress biomarkers and other MetS features were positively related with higher TAC consumption in patients suffering from MetS [63�65]. The macronutrient distribution set in a weight loss dietary plan has commonly been 50%�55% total caloric value from CHO, 15% from proteins and 30% from lipids [57,68]. However, as most people have difficulty in maintaining weight loss achievements over time [69,70], research on increment of protein intake (>20%) at the expense of CHO was carried out [71�77].
The macronutrient distribution set in a weight loss dietary plan has commonly been 50%�55% total caloric value from CHO, 15% from proteins and 30% from lipids [57,68]. However, as most people have difficulty in maintaining weight loss achievements over time [69,70], research on increment of protein intake (>20%) at the expense of CHO was carried out [71�77].
 The concept of the
The concept of the  New studies focused on the molecular action of nutritional bioactive compounds with positive effects on MetS are currently an objective of scientific research worldwide with the aim of designing more personalized strategies in the framework of molecular nutrition. Among them, flavonoids and antioxidant vitamins are some of the most studied compounds with different potential benefits such as antioxidant, vasodilatory, anti-atherogenic, antithrombotic, and anti-inflammatory effects [119]. Table 3 summarizes different nutritional bioactive compounds with potential positive effects on MetS, including the possible molecular mechanism of action involved.
New studies focused on the molecular action of nutritional bioactive compounds with positive effects on MetS are currently an objective of scientific research worldwide with the aim of designing more personalized strategies in the framework of molecular nutrition. Among them, flavonoids and antioxidant vitamins are some of the most studied compounds with different potential benefits such as antioxidant, vasodilatory, anti-atherogenic, antithrombotic, and anti-inflammatory effects [119]. Table 3 summarizes different nutritional bioactive compounds with potential positive effects on MetS, including the possible molecular mechanism of action involved.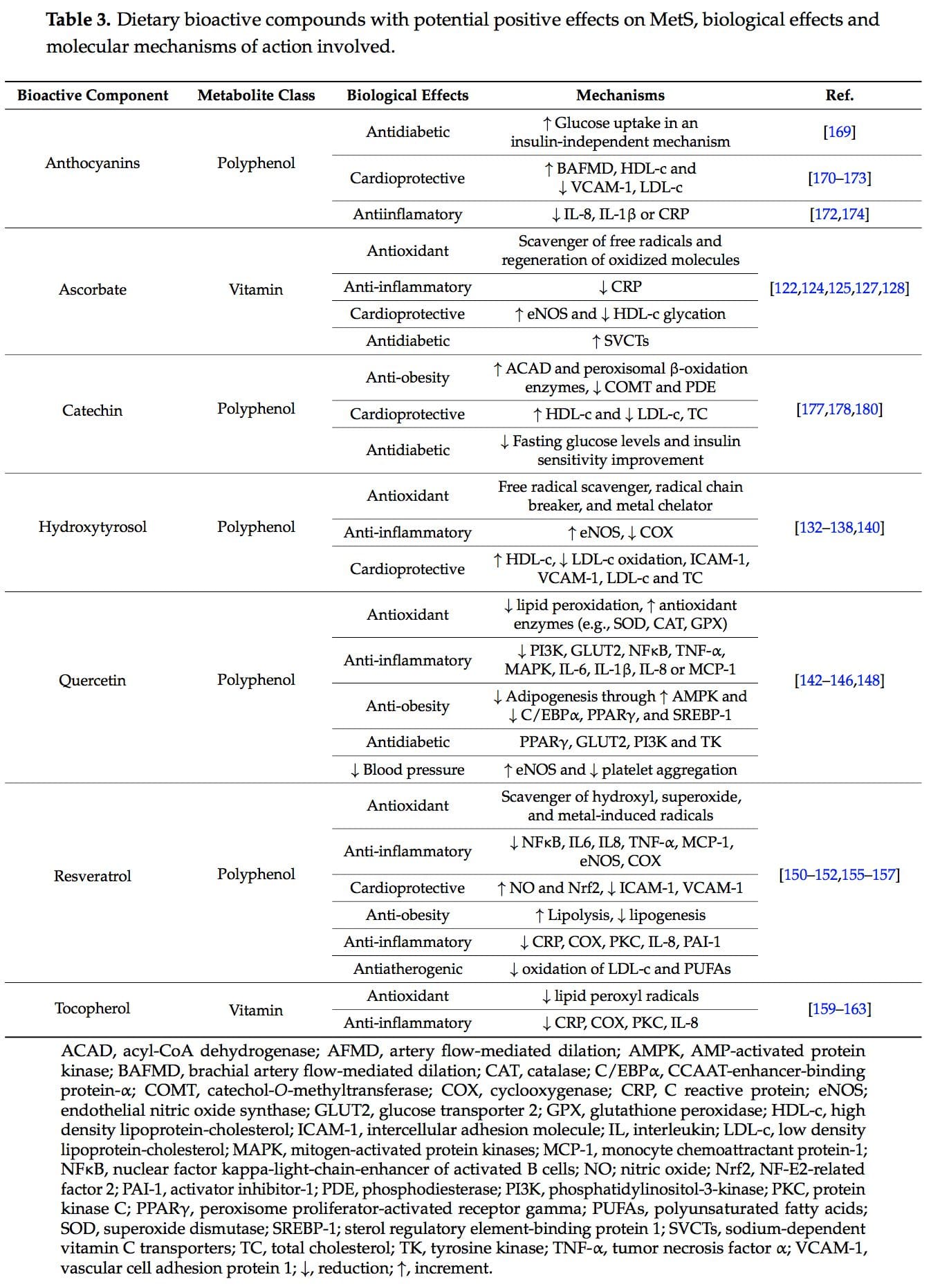
 Vitamin C, ascorbic acid or ascorbate is an essential nutrient as human beings cannot synthesize it. It is a water-soluble antioxidant mainly found in fruits, especially citrus (lemon, orange), and vegetables (pepper, kale) [120]. Several beneficial effects have been associated to this vitamin such as antioxidant and anti-inflammatory properties and prevention or treatment of CVD and type 2 diabetes [121�123].
Vitamin C, ascorbic acid or ascorbate is an essential nutrient as human beings cannot synthesize it. It is a water-soluble antioxidant mainly found in fruits, especially citrus (lemon, orange), and vegetables (pepper, kale) [120]. Several beneficial effects have been associated to this vitamin such as antioxidant and anti-inflammatory properties and prevention or treatment of CVD and type 2 diabetes [121�123]. Hydroxytyrosol (3,4-dihydroxyphenylethanol) is a phenolic compound mainly found in olives [132].
Hydroxytyrosol (3,4-dihydroxyphenylethanol) is a phenolic compound mainly found in olives [132]. Quercetin is a predominant flavanol naturally present in vegetables, fruits, green tea or red wine. It is commonly found as glycoside forms, where rutin is the most common and important structure found in nature [141].
Quercetin is a predominant flavanol naturally present in vegetables, fruits, green tea or red wine. It is commonly found as glycoside forms, where rutin is the most common and important structure found in nature [141].
 Tocopherols, also known as vitamin E, are a family of eight fat-soluble phenolic compounds whose main dietary sources are vegetable oils, nuts and seeds [130,158].
Tocopherols, also known as vitamin E, are a family of eight fat-soluble phenolic compounds whose main dietary sources are vegetable oils, nuts and seeds [130,158].
 Catechins are polyphenols that can be found in a variety of foods including fruits, vegetables, chocolate, wine, and tea [175]. The epigallocatechin 3-gallate present in tea leaves is the catechin class most studied [176].
Catechins are polyphenols that can be found in a variety of foods including fruits, vegetables, chocolate, wine, and tea [175]. The epigallocatechin 3-gallate present in tea leaves is the catechin class most studied [176].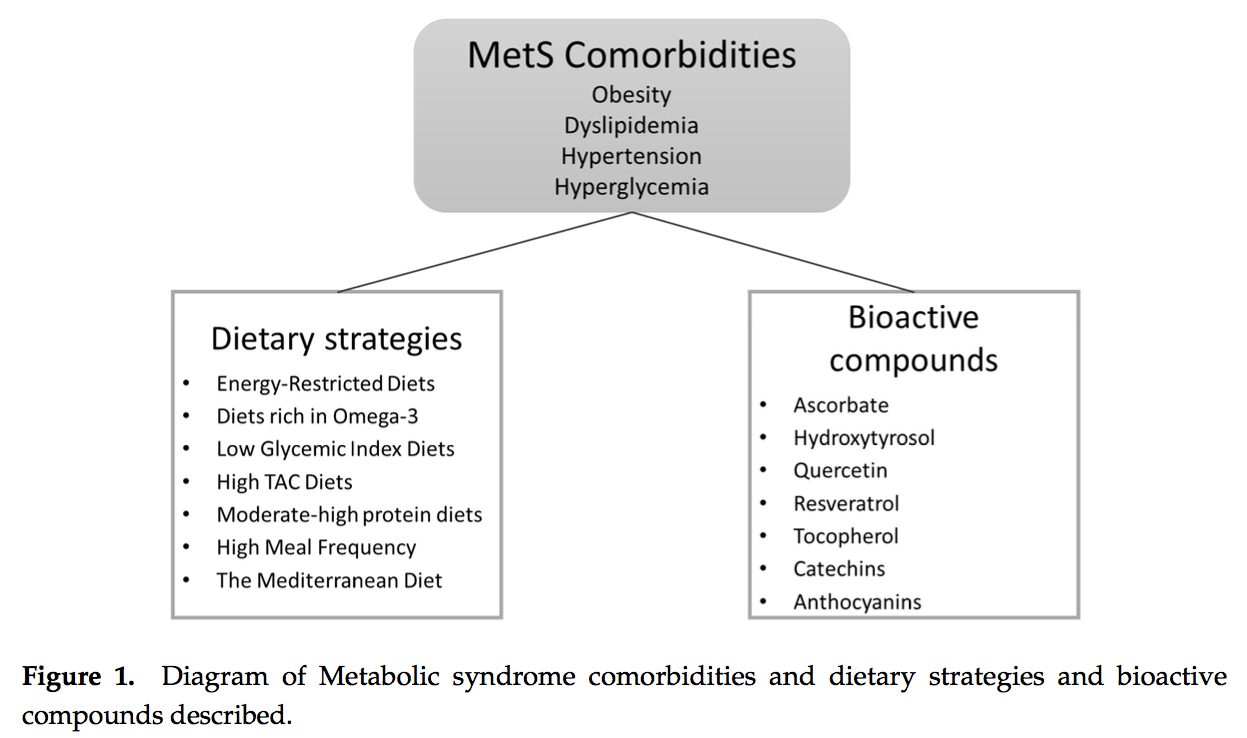


 PubMed was searched from the earliest possible date to May 2014 to identify review articles that outlined the pathophysiology of MetS and T2DM. This led to further search refinements to identify inflammatory mechanisms that occur in the pancreas, adipose tissue, skeletal muscle, and hypothalamus. Searches were also refined to identify relationships among diet, supplements, and glycemic regulation. Both animal and human studies were reviewed. The selection of specific supplements was based on those that were most commonly used in the clinical setting, namely, gymnema sylvestre, vanadium, chromium and ?-lipoic acid.
PubMed was searched from the earliest possible date to May 2014 to identify review articles that outlined the pathophysiology of MetS and T2DM. This led to further search refinements to identify inflammatory mechanisms that occur in the pancreas, adipose tissue, skeletal muscle, and hypothalamus. Searches were also refined to identify relationships among diet, supplements, and glycemic regulation. Both animal and human studies were reviewed. The selection of specific supplements was based on those that were most commonly used in the clinical setting, namely, gymnema sylvestre, vanadium, chromium and ?-lipoic acid.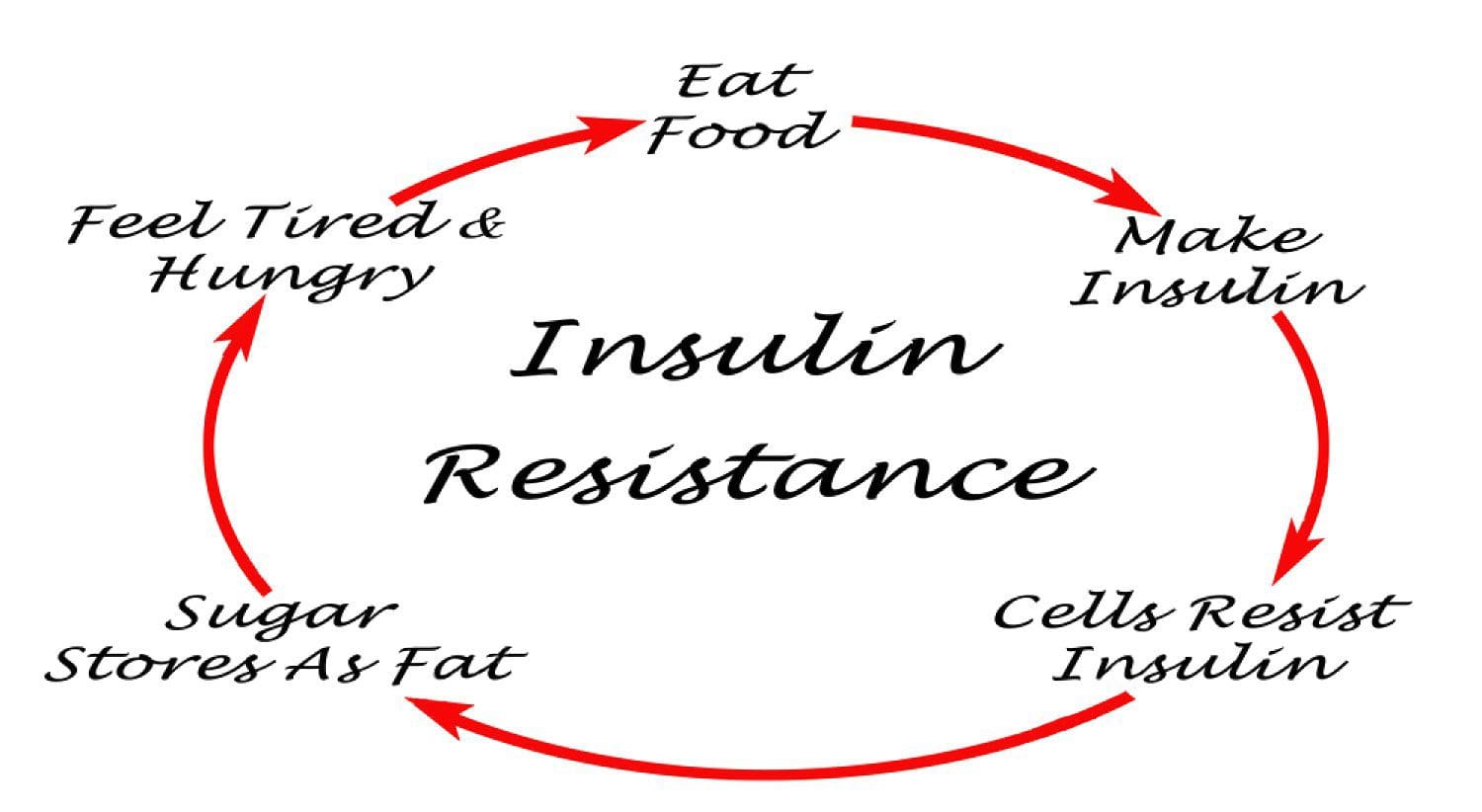 Under normal conditions, skeletal muscle, hepatic, and adipose tissues require the action of insulin for cellular glucose entry. Insulin resistance represents an inability of insulin to signal glucose passage into insulin-dependent cells. Although a genetic predisposition can exist, the�etiology of insulin resistance has been linked to chronic low-grade inflammation.1 Combined with insulin resistance-induced hyperglycemia, chronic low-grade inflammation also sustains MetS pathophysiology.1
Under normal conditions, skeletal muscle, hepatic, and adipose tissues require the action of insulin for cellular glucose entry. Insulin resistance represents an inability of insulin to signal glucose passage into insulin-dependent cells. Although a genetic predisposition can exist, the�etiology of insulin resistance has been linked to chronic low-grade inflammation.1 Combined with insulin resistance-induced hyperglycemia, chronic low-grade inflammation also sustains MetS pathophysiology.1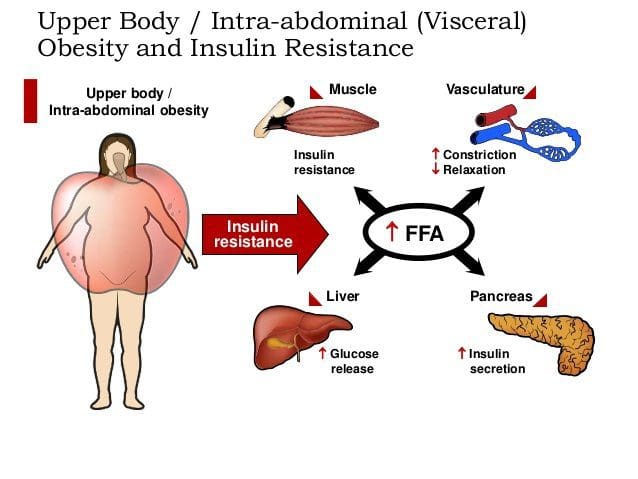 Caloric excess and a sedentary lifestyle contribute to the accumulation of subcutaneous and visceral adipose tissue. Adipose tissue was once thought of as a metabolically inert passive energy depot. A large body of evidence now demonstrates that excess visceral adipose tissue acts as a driver of chronic low-grade inflammation and insulin resistance.27,34
Caloric excess and a sedentary lifestyle contribute to the accumulation of subcutaneous and visceral adipose tissue. Adipose tissue was once thought of as a metabolically inert passive energy depot. A large body of evidence now demonstrates that excess visceral adipose tissue acts as a driver of chronic low-grade inflammation and insulin resistance.27,34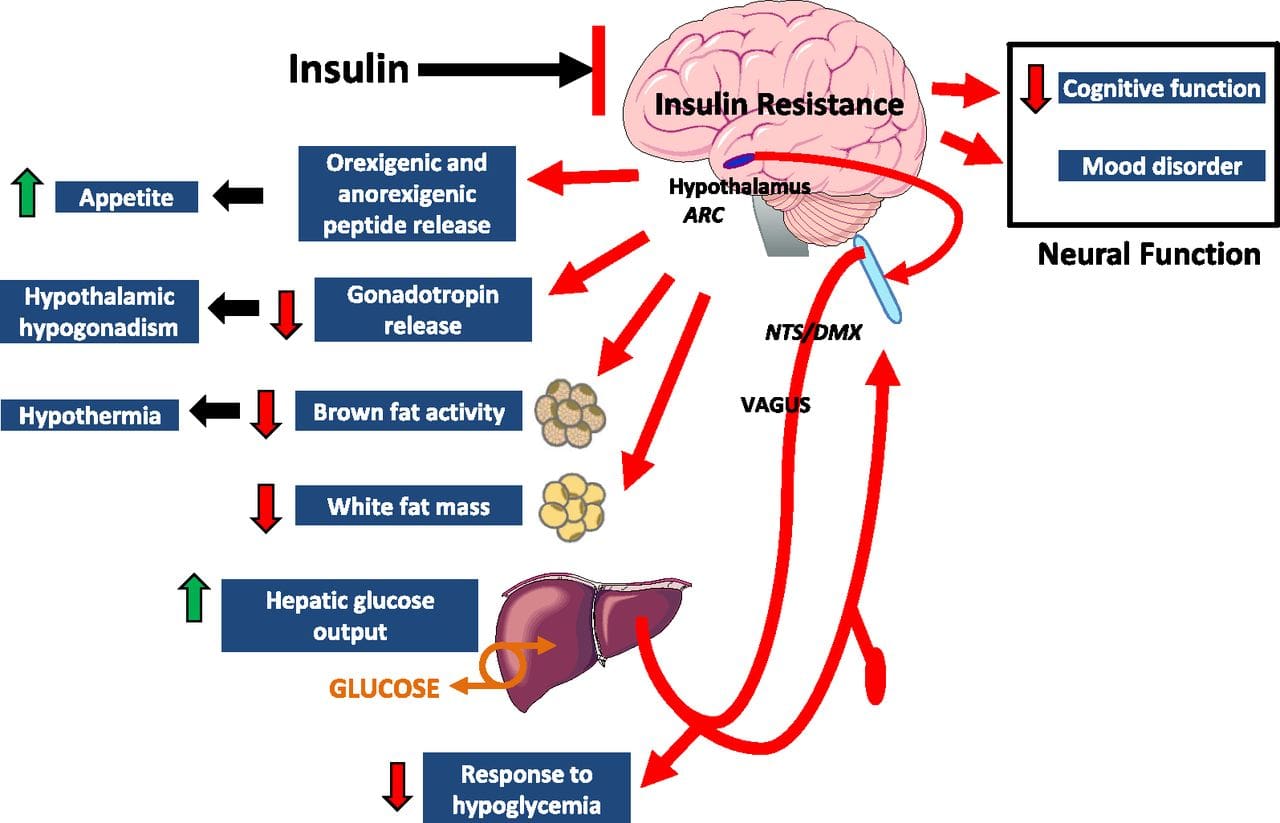 Eating behavior in the obese and overweight has been popularly attributed to a lack of will power or genetics. However, recent research has demonstrated a link between hypothalamic inflammation and increased body weight.41,41
Eating behavior in the obese and overweight has been popularly attributed to a lack of will power or genetics. However, recent research has demonstrated a link between hypothalamic inflammation and increased body weight.41,41 Feeding generally leads to a short-term increase in both oxidative stress and inflammation. 41 Total�calories consumed, glycemic index, and fatty acid profile of a meal all influence the degree of postprandial inflammation. It is estimated that the average American consumes approximately 20% of calories from refined sugar, 20% from refined grains and flour, 15% to 20% from excessively fatty meat products, and 20% from refined seed/legume oils.45 This pattern of eating contains a macronutrient composition and glycemic index that promote hyperglycemia, hyperlipemia, and an acute postprandial inflammatory response. 46 Collectively referred to as postprandial dysmetabolism, this pro-inflammatory response can sustain levels of chronic low-grade inflammation that leads to excess body fat, coronary heart disease (CHD), insulin resistance, and T2DM.28,29,47
Feeding generally leads to a short-term increase in both oxidative stress and inflammation. 41 Total�calories consumed, glycemic index, and fatty acid profile of a meal all influence the degree of postprandial inflammation. It is estimated that the average American consumes approximately 20% of calories from refined sugar, 20% from refined grains and flour, 15% to 20% from excessively fatty meat products, and 20% from refined seed/legume oils.45 This pattern of eating contains a macronutrient composition and glycemic index that promote hyperglycemia, hyperlipemia, and an acute postprandial inflammatory response. 46 Collectively referred to as postprandial dysmetabolism, this pro-inflammatory response can sustain levels of chronic low-grade inflammation that leads to excess body fat, coronary heart disease (CHD), insulin resistance, and T2DM.28,29,47 Diagnosis of MetS has been linked to an increased risk of developing T2DM and cardiovascular disease over the following 5 to 10 years. 1 It further increases a patient’s risk of stroke, myocardial infarction, and death from any of the aforementioned conditions.1
Diagnosis of MetS has been linked to an increased risk of developing T2DM and cardiovascular disease over the following 5 to 10 years. 1 It further increases a patient’s risk of stroke, myocardial infarction, and death from any of the aforementioned conditions.1 Research has identified nutrients that play key roles in promoting proper insulin sensitivity, including vitamin D, magnesium, omega-3 (n-3) fatty acids, curcumin, gymnema, vanadium, chromium, and ?-lipoic acid. It is possible to get adequate vitamin D from sun exposure and adequate amounts of magnesium and omega-3 fatty acids from food. Contrastingly, the therapeutic levels of chromium and ?-lipoic acid that affect insulin sensitivity and reduce�insulin resistance cannot be obtained in food and must be supplemented.
Research has identified nutrients that play key roles in promoting proper insulin sensitivity, including vitamin D, magnesium, omega-3 (n-3) fatty acids, curcumin, gymnema, vanadium, chromium, and ?-lipoic acid. It is possible to get adequate vitamin D from sun exposure and adequate amounts of magnesium and omega-3 fatty acids from food. Contrastingly, the therapeutic levels of chromium and ?-lipoic acid that affect insulin sensitivity and reduce�insulin resistance cannot be obtained in food and must be supplemented. Vitamin D, magnesium, and n-3 fatty acids have multiple functions, and generalized inflammation reduction is a common mechanism of action.74�80 Their supplemental use should be considered in the context of low-grade inflammation reduction and health promotion, rather than as a specific treatment for MetS or T2DM.
Vitamin D, magnesium, and n-3 fatty acids have multiple functions, and generalized inflammation reduction is a common mechanism of action.74�80 Their supplemental use should be considered in the context of low-grade inflammation reduction and health promotion, rather than as a specific treatment for MetS or T2DM. Gymnemic acids are the active component of the G sylvestre plant leaves. Gymnemic acids are the active component of the G sylvestre plant leaves. Studies evaluating G sylvestre’s effects on diabetes in humans have generally been of poor methodological quality. Experimental animal studies have found that gymnemic acids may decrease glucose uptake in the small intestine, inhibit gluconeogenesis, and reduce hepatic and skeletal muscle insulin resistance.99 Other animal studies suggest that gymnemic acids may have comparable efficacy in reducing blood sugar levels to the first-generation sulfonylurea, tolbutamide.100
Gymnemic acids are the active component of the G sylvestre plant leaves. Gymnemic acids are the active component of the G sylvestre plant leaves. Studies evaluating G sylvestre’s effects on diabetes in humans have generally been of poor methodological quality. Experimental animal studies have found that gymnemic acids may decrease glucose uptake in the small intestine, inhibit gluconeogenesis, and reduce hepatic and skeletal muscle insulin resistance.99 Other animal studies suggest that gymnemic acids may have comparable efficacy in reducing blood sugar levels to the first-generation sulfonylurea, tolbutamide.100 Vanadyl sulfate has been reported to prolong the events of insulin signaling and may actually improve insulin sensitivity.101 Limited data suggest that it inhibits gluconeogenesis, possibly ameliorating hepatic insulin resistance. 100,101 Uncontrolled clinical trials have reported improvements in insulin sensitivity using 50 to 300 mg daily for periods ranging from 3 to 6 weeks. 101�103 Contrastingly, a recent randomized, double-blind, placebo-controlled trial found that 50 mg of vanadyl sulfate twice daily for 4 weeks had no effect in individuals with impaired glucose tolerance. 104 Limited clinical and experimental data exist supporting the use of vanadyl sulfate to improve insulin resistance,�and further research is warranted regarding its safety and efficacy.
Vanadyl sulfate has been reported to prolong the events of insulin signaling and may actually improve insulin sensitivity.101 Limited data suggest that it inhibits gluconeogenesis, possibly ameliorating hepatic insulin resistance. 100,101 Uncontrolled clinical trials have reported improvements in insulin sensitivity using 50 to 300 mg daily for periods ranging from 3 to 6 weeks. 101�103 Contrastingly, a recent randomized, double-blind, placebo-controlled trial found that 50 mg of vanadyl sulfate twice daily for 4 weeks had no effect in individuals with impaired glucose tolerance. 104 Limited clinical and experimental data exist supporting the use of vanadyl sulfate to improve insulin resistance,�and further research is warranted regarding its safety and efficacy. Diets high in refined sugar and flour are deficient in chromium (Cr) and lead to an increased urinary excretion of chromium. 105,106 The progression of MetS is not likely caused by a chromium deficiency, 107 and dosages that benefit glycemic regulation are not achievable through food. 106,108,109
Diets high in refined sugar and flour are deficient in chromium (Cr) and lead to an increased urinary excretion of chromium. 105,106 The progression of MetS is not likely caused by a chromium deficiency, 107 and dosages that benefit glycemic regulation are not achievable through food. 106,108,109 Humans derive ?-lipoic acid through dietary means and from endogenous synthesis. 111 The foods richest in ?-lipoic acid are animal tissues with extensive metabolic activity such as animal heart, liver, and kidney, which are not consumed in large amounts in the typical American diet. 111 Supplemental amounts of ?-lipoic acid used in the treatment of T2DM (300-600 mg) are likely to be as much as 1000 times greater than the amounts that could be obtained from the diet.112
Humans derive ?-lipoic acid through dietary means and from endogenous synthesis. 111 The foods richest in ?-lipoic acid are animal tissues with extensive metabolic activity such as animal heart, liver, and kidney, which are not consumed in large amounts in the typical American diet. 111 Supplemental amounts of ?-lipoic acid used in the treatment of T2DM (300-600 mg) are likely to be as much as 1000 times greater than the amounts that could be obtained from the diet.112 This is a narrative overview of the topic of MetS. A systematic review was not performed; therefore, there may be relevant information missing from this review. The contents of this overview focuses on the opinions of the authors, and therefore, others may disagree with our opinions or approaches to management. This overview is limited by the studies that have been published. To date, no studies have been published that identify the effectiveness of a combination of a dietary intervention, such as the Spanish
This is a narrative overview of the topic of MetS. A systematic review was not performed; therefore, there may be relevant information missing from this review. The contents of this overview focuses on the opinions of the authors, and therefore, others may disagree with our opinions or approaches to management. This overview is limited by the studies that have been published. To date, no studies have been published that identify the effectiveness of a combination of a dietary intervention, such as the Spanish 
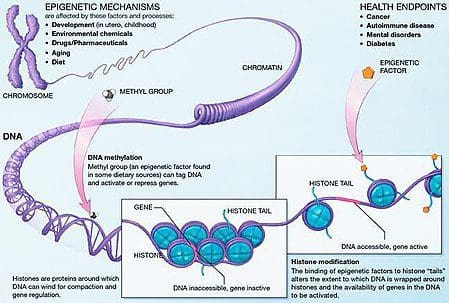 Obesity is a complex, multifactorial disease, and better understanding of the mechanisms underlying the interactions between lifestyle, environment, and genetics is critical for developing effective strategies for prevention and treatment [1].
Obesity is a complex, multifactorial disease, and better understanding of the mechanisms underlying the interactions between lifestyle, environment, and genetics is critical for developing effective strategies for prevention and treatment [1]. Animal models provide unique opportunities for highly controlled studies that provide mechanistic insight into�the role of specific epigenetic marks, both as indicators of current metabolic status and as predictors of the future risk of obesity and metabolic disease. A particularly important aspect of animal studies is that they allow for the assessment of epigenetic changes within target tissues, including the liver and hypothalamus, which is much more difficult in humans. Moreover, the ability to harvest large quantities of fresh tissue makes it possible to assess multiple chromatin marks as well as DNA methylation. Some of these epigenetic modifications either alone or in combination may be responsive to environmental programming. In animal models, it is also possible to study multiple generations of offspring and thus enable differentiation between trans-generational and intergenerational transmission of obesity risk mediated by epigenetic memory of parental nutritional status, which cannot be easily distinguished in human studies. We use the former term for meiotic transmission of risk in the absence of continued exposure while the latter primarily entails direct transmission of risk through metabolic reprogramming of the fetus or gametes.
Animal models provide unique opportunities for highly controlled studies that provide mechanistic insight into�the role of specific epigenetic marks, both as indicators of current metabolic status and as predictors of the future risk of obesity and metabolic disease. A particularly important aspect of animal studies is that they allow for the assessment of epigenetic changes within target tissues, including the liver and hypothalamus, which is much more difficult in humans. Moreover, the ability to harvest large quantities of fresh tissue makes it possible to assess multiple chromatin marks as well as DNA methylation. Some of these epigenetic modifications either alone or in combination may be responsive to environmental programming. In animal models, it is also possible to study multiple generations of offspring and thus enable differentiation between trans-generational and intergenerational transmission of obesity risk mediated by epigenetic memory of parental nutritional status, which cannot be easily distinguished in human studies. We use the former term for meiotic transmission of risk in the absence of continued exposure while the latter primarily entails direct transmission of risk through metabolic reprogramming of the fetus or gametes.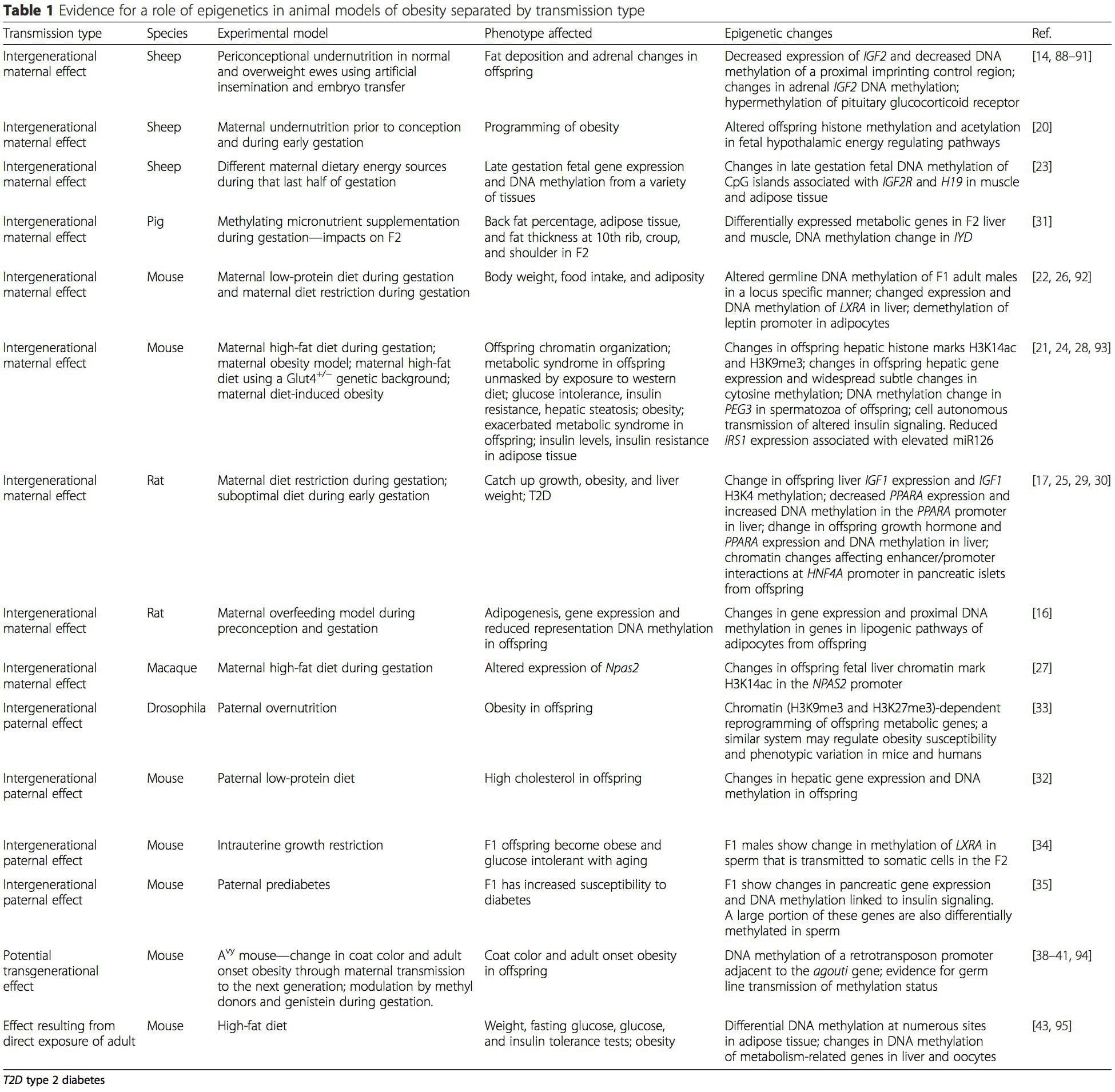 (i) Epigenetic Changes In Offspring Associated With Maternal Nutrition During Gestation
(i) Epigenetic Changes In Offspring Associated With Maternal Nutrition During Gestation Maternal nutritional supplementation, undernutrition, and over nutrition during pregnancy can alter fat deposition and energy homeostasis in offspring [11, 13�15, 19]. Associated with these effects in the offspring are changes in DNA methylation, histone post-translational modifications, and gene expression for several target genes,�especially genes regulating fatty acid metabolism and insulin signaling [16, 17, 20�30]. The diversity of animal models used in these studies and the common metabolic pathways impacted suggest an evolutionarily conserved adaptive response mediated by epigenetic modification. However, few of the specific identified genes and epigenetic changes have been cross-validated in related studies, and large-scale genome-wide investigations have typically not been applied. A major hindrance to comparison of these studies is the different develop mental windows subjected to nutritional challenge, which may cause considerably different outcomes. Proof that the epigenetic changes are causal rather than being associated with offspring phenotypic changes is also required. This will necessitate the identification of a parental nutritionally induced epigenetic �memory� response that precedes development of the altered phenotype in offspring.
Maternal nutritional supplementation, undernutrition, and over nutrition during pregnancy can alter fat deposition and energy homeostasis in offspring [11, 13�15, 19]. Associated with these effects in the offspring are changes in DNA methylation, histone post-translational modifications, and gene expression for several target genes,�especially genes regulating fatty acid metabolism and insulin signaling [16, 17, 20�30]. The diversity of animal models used in these studies and the common metabolic pathways impacted suggest an evolutionarily conserved adaptive response mediated by epigenetic modification. However, few of the specific identified genes and epigenetic changes have been cross-validated in related studies, and large-scale genome-wide investigations have typically not been applied. A major hindrance to comparison of these studies is the different develop mental windows subjected to nutritional challenge, which may cause considerably different outcomes. Proof that the epigenetic changes are causal rather than being associated with offspring phenotypic changes is also required. This will necessitate the identification of a parental nutritionally induced epigenetic �memory� response that precedes development of the altered phenotype in offspring. Emerging studies have demonstrated that paternal plane of nutrition can impact offspring fat deposition and epigenetic marks [31�34]. One recent investigation using mice has demonstrated that paternal pre-diabetes leads to increased susceptibility to diabetes in F1 offspring with associated changes in pancreatic gene expression and DNA methylation linked to insulin signaling [35]. Importantly, there was an overlap of these epigenetic changes in pancreatic islets and sperm suggesting germ line inheritance. However, most of these studies, although intriguing in their implications, are limited in the genomic scale of investigation and frequently show weak and somewhat transient epigenetic alterations associated with mild metabolic phenotypes in offspring.
Emerging studies have demonstrated that paternal plane of nutrition can impact offspring fat deposition and epigenetic marks [31�34]. One recent investigation using mice has demonstrated that paternal pre-diabetes leads to increased susceptibility to diabetes in F1 offspring with associated changes in pancreatic gene expression and DNA methylation linked to insulin signaling [35]. Importantly, there was an overlap of these epigenetic changes in pancreatic islets and sperm suggesting germ line inheritance. However, most of these studies, although intriguing in their implications, are limited in the genomic scale of investigation and frequently show weak and somewhat transient epigenetic alterations associated with mild metabolic phenotypes in offspring.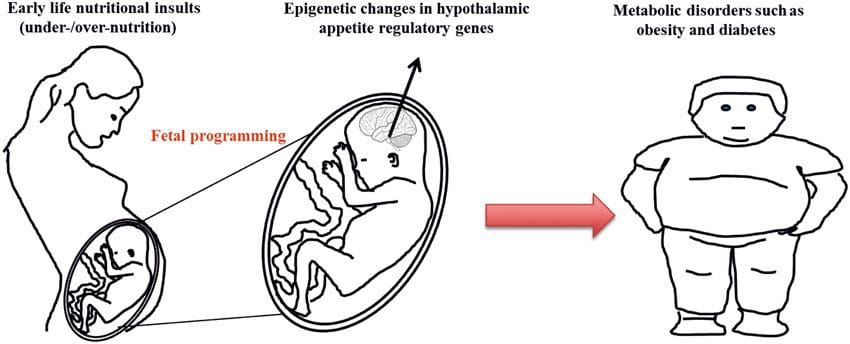 Stable transmission of epigenetic information across multiple generations is well described in plant systems and C. elegans, but its significance in mammals is still much debated [36, 37]. An epigenetic basis for grand- parental transmission of phenotypes in response to dietary exposures has been well established, including in livestock species [31]. The most influential studies demonstrating effects of epigenetic transmission impacting offspring phenotype have used the example of the viable yellow agouti (Avy) mouse [38]. In this mouse, an insertion of a retrotransposon upstream of the agouti gene causes its constitutive expression and consequent yellow coat color and adult onset obesity. Maternal transmission through the germ line results in DNA methylation�mediated silencing of agouti expression resulting in wild-type coat color and lean phenotype of the offspring [39, 40]. Importantly, subsequent studies in these mice demonstrated that maternal exposure to methyl donors causes a shift in coat color [41]. One study has reported transmission of a phenotype to the F3 generation and alterations in expression of large number of genes in response to protein restriction in F0 [42]; however, alterations in expression were highly variable and a direct link to epigenetic changes was not identified in this system.
Stable transmission of epigenetic information across multiple generations is well described in plant systems and C. elegans, but its significance in mammals is still much debated [36, 37]. An epigenetic basis for grand- parental transmission of phenotypes in response to dietary exposures has been well established, including in livestock species [31]. The most influential studies demonstrating effects of epigenetic transmission impacting offspring phenotype have used the example of the viable yellow agouti (Avy) mouse [38]. In this mouse, an insertion of a retrotransposon upstream of the agouti gene causes its constitutive expression and consequent yellow coat color and adult onset obesity. Maternal transmission through the germ line results in DNA methylation�mediated silencing of agouti expression resulting in wild-type coat color and lean phenotype of the offspring [39, 40]. Importantly, subsequent studies in these mice demonstrated that maternal exposure to methyl donors causes a shift in coat color [41]. One study has reported transmission of a phenotype to the F3 generation and alterations in expression of large number of genes in response to protein restriction in F0 [42]; however, alterations in expression were highly variable and a direct link to epigenetic changes was not identified in this system.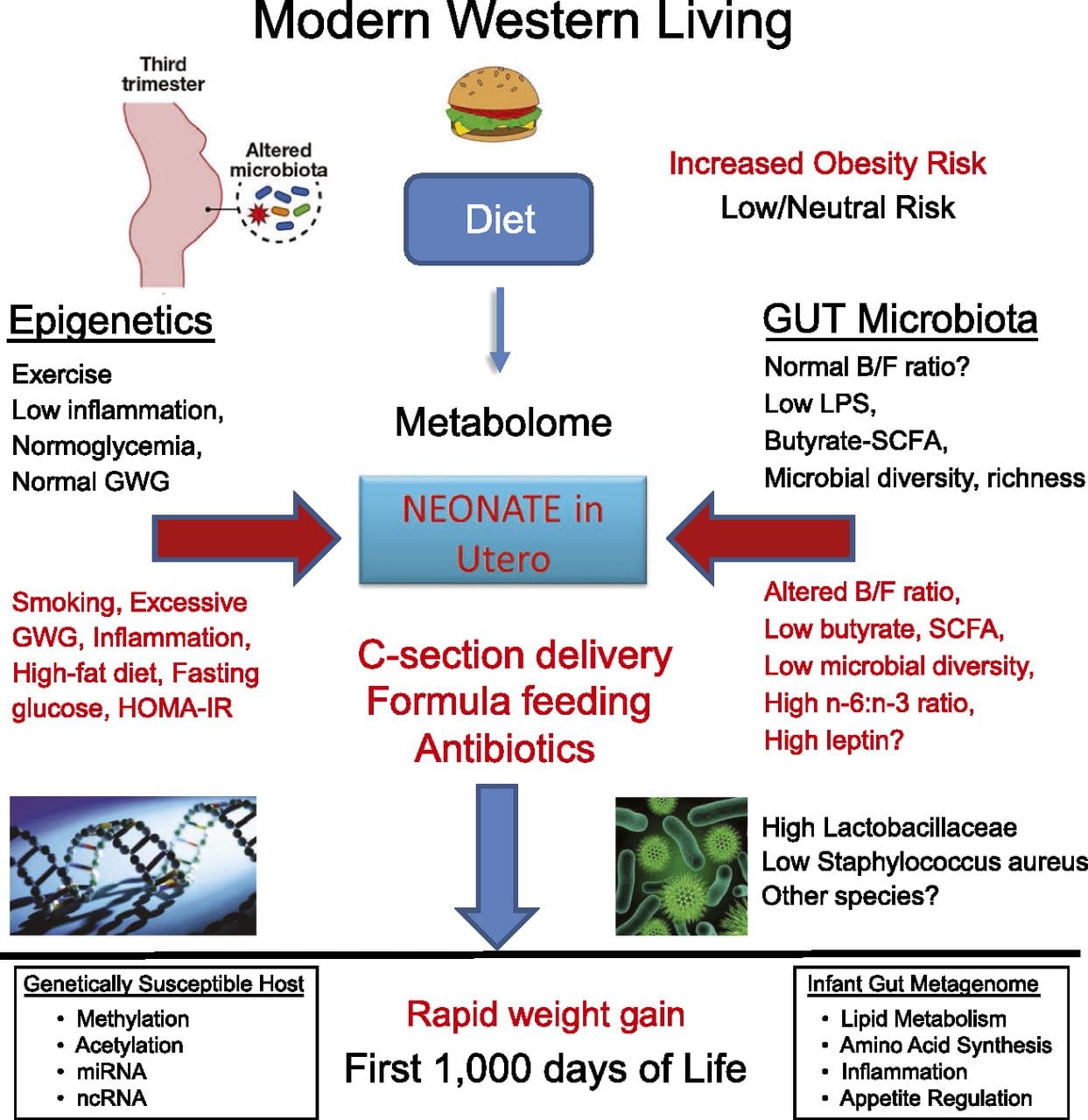 While many studies have identified diet-associated epigenetic changes in animal models using candidate site-specific regions, there have been few genome-wide analyses undertaken. A recent study focussed on determining the direct epigenetic impact of high-fat diets/ diet-induced obesity in adult mice using genome-wide gene expression and DNA methylation analyses [43]. This study identified 232 differentially methylated regions (DMRs) in adipocytes from control and high-fat fed mice. Importantly, the corresponding human regions for the murine DMRs were also differentially methylated in adipose tissue from a population of obese and lean humans, thereby highlighting the remarkable evolutionary conservation of these regions. This result emphasizes the likely importance of the identified DMRs in regulating energy homeostasis in mammals.
While many studies have identified diet-associated epigenetic changes in animal models using candidate site-specific regions, there have been few genome-wide analyses undertaken. A recent study focussed on determining the direct epigenetic impact of high-fat diets/ diet-induced obesity in adult mice using genome-wide gene expression and DNA methylation analyses [43]. This study identified 232 differentially methylated regions (DMRs) in adipocytes from control and high-fat fed mice. Importantly, the corresponding human regions for the murine DMRs were also differentially methylated in adipose tissue from a population of obese and lean humans, thereby highlighting the remarkable evolutionary conservation of these regions. This result emphasizes the likely importance of the identified DMRs in regulating energy homeostasis in mammals.
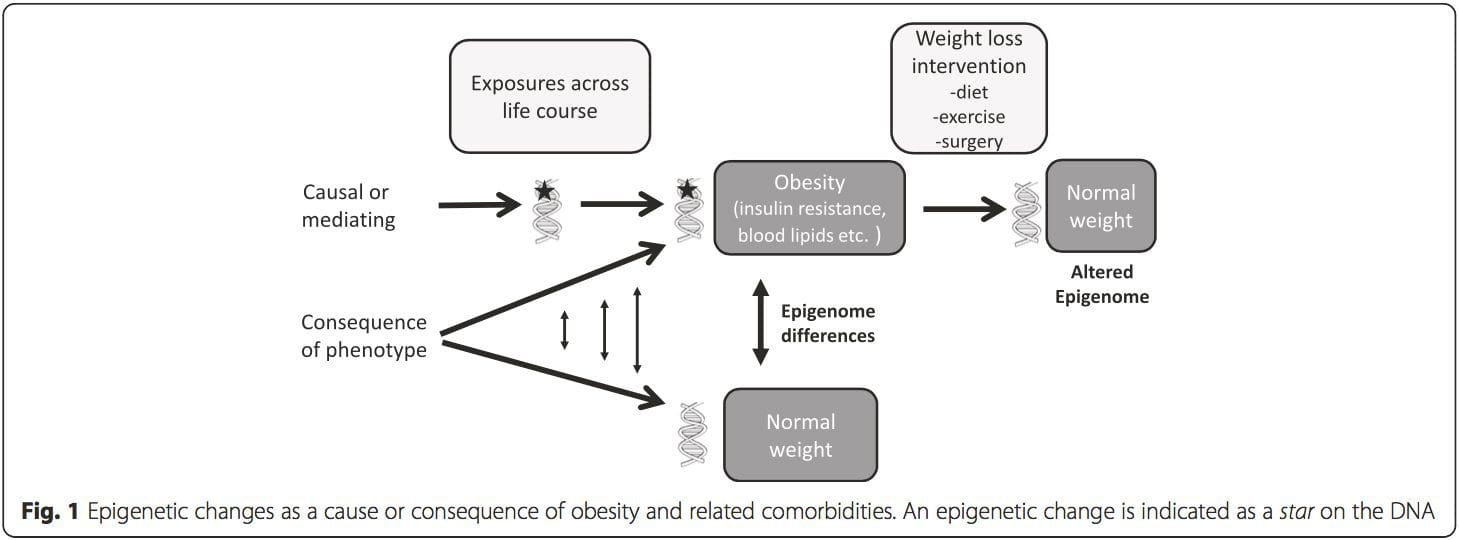 (i) Genetic association studies. Genetic polymorphisms that are associated with an increased risk of developing particular conditions are a priori linked to the causative genes. The presence of differential�methylation in such regions infers functional relevance of these epigenetic changes in controlling expression of the proximal gene(s). There are strong cis-acting genetic effects underpinning much epigenetic variation [7, 45], and in population-based studies, methods that use genetic surrogates to infer a causal or mediating role of epigenome differences have been applied [7, 46�48]. The use of familial genetic information can also lead to the identification of potentially causative candidate regions showing phenotype-related differential methylation [49].
(i) Genetic association studies. Genetic polymorphisms that are associated with an increased risk of developing particular conditions are a priori linked to the causative genes. The presence of differential�methylation in such regions infers functional relevance of these epigenetic changes in controlling expression of the proximal gene(s). There are strong cis-acting genetic effects underpinning much epigenetic variation [7, 45], and in population-based studies, methods that use genetic surrogates to infer a causal or mediating role of epigenome differences have been applied [7, 46�48]. The use of familial genetic information can also lead to the identification of potentially causative candidate regions showing phenotype-related differential methylation [49].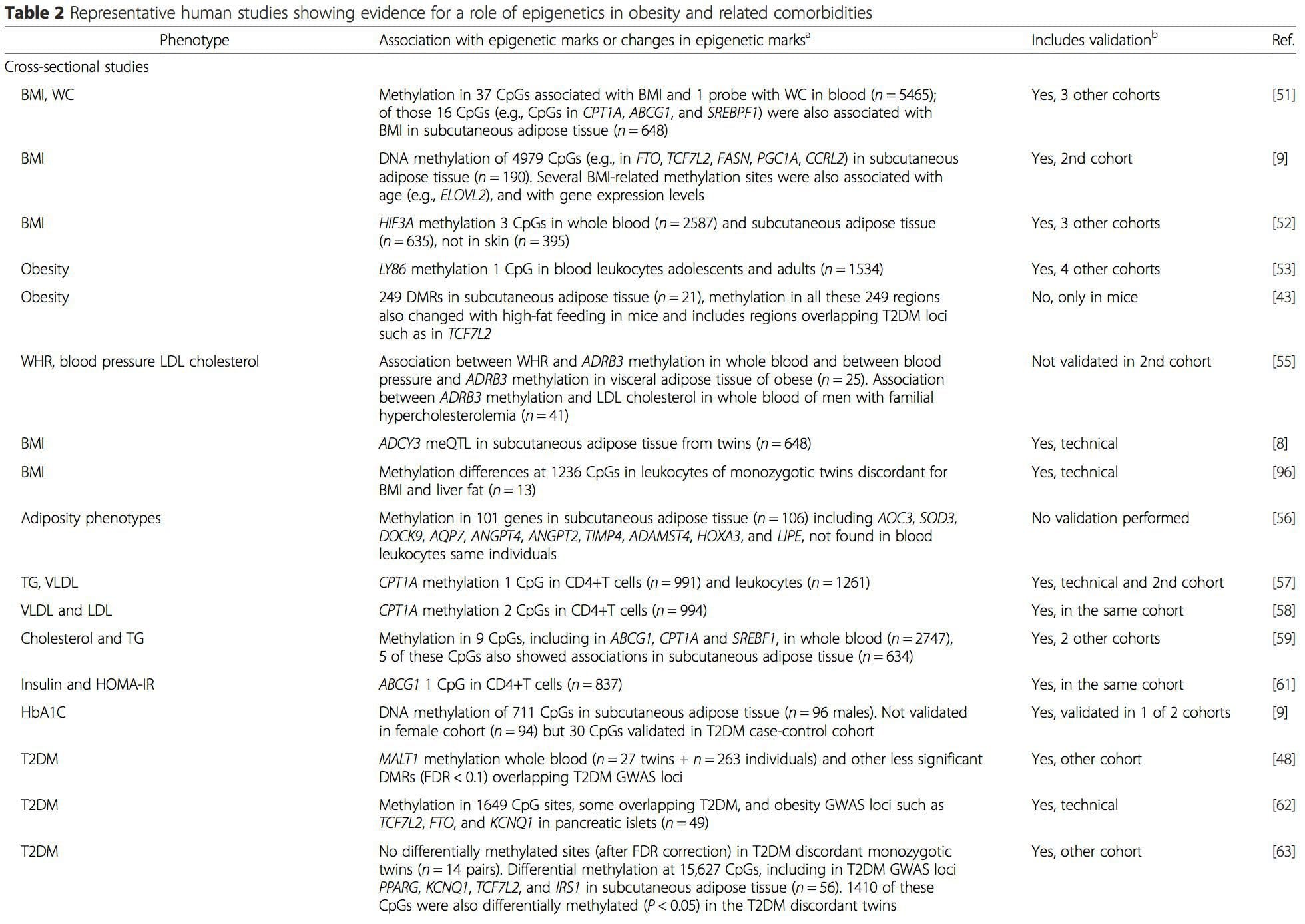
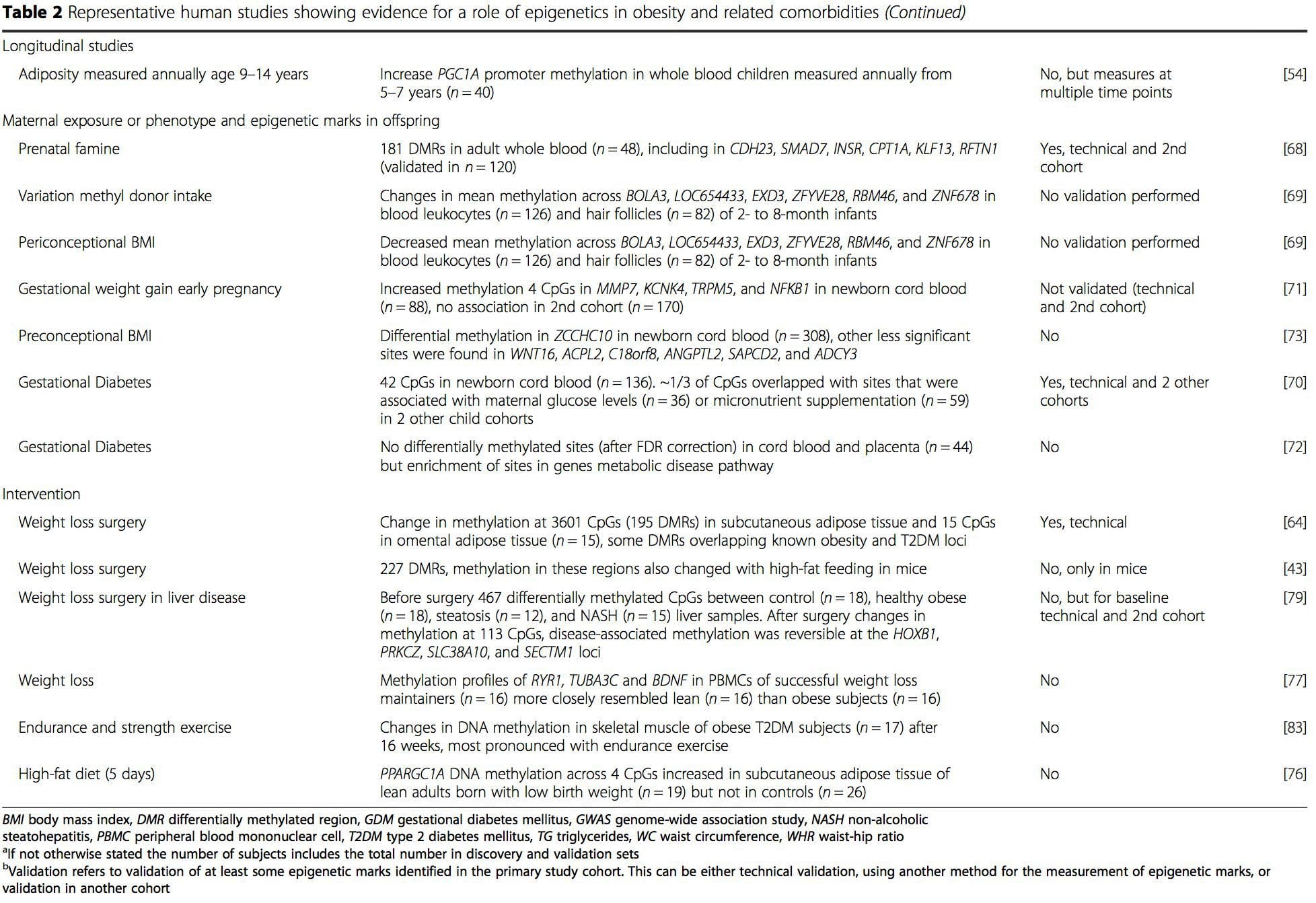 From these studies, altered methylation of PGC1A, HIF3A, ABCG1, and CPT1A and the previously described RXRA [18] have emerged as biomarkers associated with, or perhaps predictive of, metabolic health that are also plausible candidates for a role in development of metabolic disease.
From these studies, altered methylation of PGC1A, HIF3A, ABCG1, and CPT1A and the previously described RXRA [18] have emerged as biomarkers associated with, or perhaps predictive of, metabolic health that are also plausible candidates for a role in development of metabolic disease.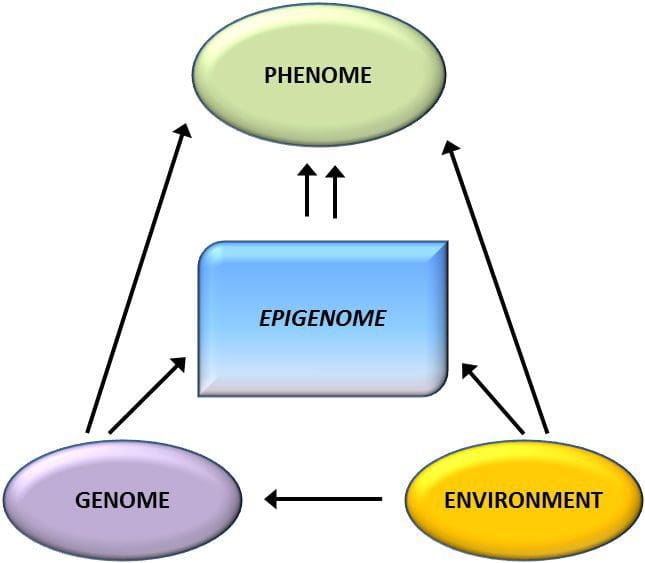 Epigenetic variation is highly influenced by the underlying genetic variation, with genotype estimated to explain ~20�40 % of the variation [6, 8]. Recently, a number of studies have begun to integrate methylome and genotype data to identify methylation quantitative trait loci (meQTL) associated with disease phenotypes. For instance, in adipose tissue, an meQTL overlapping�with a BMI genetic risk locus has been identified in an enhancer element upstream of ADCY3 [8]. Other studies have also identified overlaps between known obesity and T2DM risk loci and DMRs associated with obesity and T2DM [43, 48, 62]. Methylation of a number of such DMRs was also modulated by high-fat feeding in mice [43] and weight loss in humans [64]. These results identify an intriguing link between genetic variations linked with disease susceptibility and their association with regions of the genome that undergo epigenetic modifications in response to nutritional challenges, implying a causal relationship. The close connection between genetic and epigenetic variation may signify their essential roles in generating individual variation [65, 66]. However, while these findings suggest that DNA methylation may be a mediator of genetic effects, it is also important to consider that both genetic and epigenetic processes could act independently on the same genes. Twin studies [8, 63, 67] can provide important insights and indicate that inter-individual differences in levels of DNA methylation arise predominantly from non-shared environment and stochastic influences, minimally from shared environmental effects, but also with a significant impact of genetic variation.
Epigenetic variation is highly influenced by the underlying genetic variation, with genotype estimated to explain ~20�40 % of the variation [6, 8]. Recently, a number of studies have begun to integrate methylome and genotype data to identify methylation quantitative trait loci (meQTL) associated with disease phenotypes. For instance, in adipose tissue, an meQTL overlapping�with a BMI genetic risk locus has been identified in an enhancer element upstream of ADCY3 [8]. Other studies have also identified overlaps between known obesity and T2DM risk loci and DMRs associated with obesity and T2DM [43, 48, 62]. Methylation of a number of such DMRs was also modulated by high-fat feeding in mice [43] and weight loss in humans [64]. These results identify an intriguing link between genetic variations linked with disease susceptibility and their association with regions of the genome that undergo epigenetic modifications in response to nutritional challenges, implying a causal relationship. The close connection between genetic and epigenetic variation may signify their essential roles in generating individual variation [65, 66]. However, while these findings suggest that DNA methylation may be a mediator of genetic effects, it is also important to consider that both genetic and epigenetic processes could act independently on the same genes. Twin studies [8, 63, 67] can provide important insights and indicate that inter-individual differences in levels of DNA methylation arise predominantly from non-shared environment and stochastic influences, minimally from shared environmental effects, but also with a significant impact of genetic variation. Prenatal environment: Two recently published studies made use of human populations that experienced �natural� variations in nutrient supply to study the impact of maternal nutrition before or during pregnancy on DNA methylation in the offspring [68, 69]. The first study used a Gambian mother-child cohort to show that both seasonal variations in maternal methyl donor intake during pregnancy and maternal pre-pregnancy BMI were associated with altered methylation in the infants [69]. The second study utilized adult offspring from the Dutch Hunger Winter cohort to investigate the effect of prenatal exposure to an acute period of severe maternal undernutrition on DNA methylation of genes involved in growth and metabolism in adulthood [68]. The results highlighted the importance of the timing of the exposure in its impact on the epigenome, since significant epigenetic effects were only identified in individuals exposed to famine during early gestation. Importantly, the epigenetic changes occurred in conjunction with increased BMI; however, it was not possible to establish in this study whether these changes were present earlier in life or a consequence of the higher BMI.
Prenatal environment: Two recently published studies made use of human populations that experienced �natural� variations in nutrient supply to study the impact of maternal nutrition before or during pregnancy on DNA methylation in the offspring [68, 69]. The first study used a Gambian mother-child cohort to show that both seasonal variations in maternal methyl donor intake during pregnancy and maternal pre-pregnancy BMI were associated with altered methylation in the infants [69]. The second study utilized adult offspring from the Dutch Hunger Winter cohort to investigate the effect of prenatal exposure to an acute period of severe maternal undernutrition on DNA methylation of genes involved in growth and metabolism in adulthood [68]. The results highlighted the importance of the timing of the exposure in its impact on the epigenome, since significant epigenetic effects were only identified in individuals exposed to famine during early gestation. Importantly, the epigenetic changes occurred in conjunction with increased BMI; however, it was not possible to establish in this study whether these changes were present earlier in life or a consequence of the higher BMI. Postnatal environment: The epigenome is established de novo during embryonic development, and therefore, the prenatal environment most likely has the most significant impact on the epigenome. However, it is now clear that changes do occur in the �mature� epigenome under the influence of a range of conditions, including aging, exposure to toxins, and dietary alterations. For example, changes in DNA methylation in numerous genes in skeletal muscle and PGC1A in adipose tissue have been demonstrated in response to a high-fat diet [75, 76]. Interventions to lose body fat mass have also been associated with changes in DNA methylation. Studies have reported that the DNA methylation profiles of adipose tissue [43, 64], peripheral blood mononuclear cells [77], and muscle tissue [78] in formerly obese patients become more similar to the profiles of lean subjects following weight loss. Weight loss surgery also partially reversed non-alcoholic fatty liver disease-associated methylation changes in liver [79] and in another study led to hypomethylation of multiple obesity candidate genes, with more pronounced effects in subcutaneous compared to omental (visceral) fat [64]. Accumulating evidence suggests that exercise interventions can also influence DNA methylation. Most of these studies have been conducted in lean individuals [80�82], but one exercise study in obese T2DM subjects also demonstrated changes in DNA methylation, including in genes involved in fatty acid and glucose transport [83]. Epigenetic changes also occur with aging, and recent data suggest a role of obesity in augmenting them [9, 84, 85]. Obesity accelerated the epigenetic age of liver tissue, but in contrast to the findings described above, this effect was not reversible after weight loss [84].
Postnatal environment: The epigenome is established de novo during embryonic development, and therefore, the prenatal environment most likely has the most significant impact on the epigenome. However, it is now clear that changes do occur in the �mature� epigenome under the influence of a range of conditions, including aging, exposure to toxins, and dietary alterations. For example, changes in DNA methylation in numerous genes in skeletal muscle and PGC1A in adipose tissue have been demonstrated in response to a high-fat diet [75, 76]. Interventions to lose body fat mass have also been associated with changes in DNA methylation. Studies have reported that the DNA methylation profiles of adipose tissue [43, 64], peripheral blood mononuclear cells [77], and muscle tissue [78] in formerly obese patients become more similar to the profiles of lean subjects following weight loss. Weight loss surgery also partially reversed non-alcoholic fatty liver disease-associated methylation changes in liver [79] and in another study led to hypomethylation of multiple obesity candidate genes, with more pronounced effects in subcutaneous compared to omental (visceral) fat [64]. Accumulating evidence suggests that exercise interventions can also influence DNA methylation. Most of these studies have been conducted in lean individuals [80�82], but one exercise study in obese T2DM subjects also demonstrated changes in DNA methylation, including in genes involved in fatty acid and glucose transport [83]. Epigenetic changes also occur with aging, and recent data suggest a role of obesity in augmenting them [9, 84, 85]. Obesity accelerated the epigenetic age of liver tissue, but in contrast to the findings described above, this effect was not reversible after weight loss [84].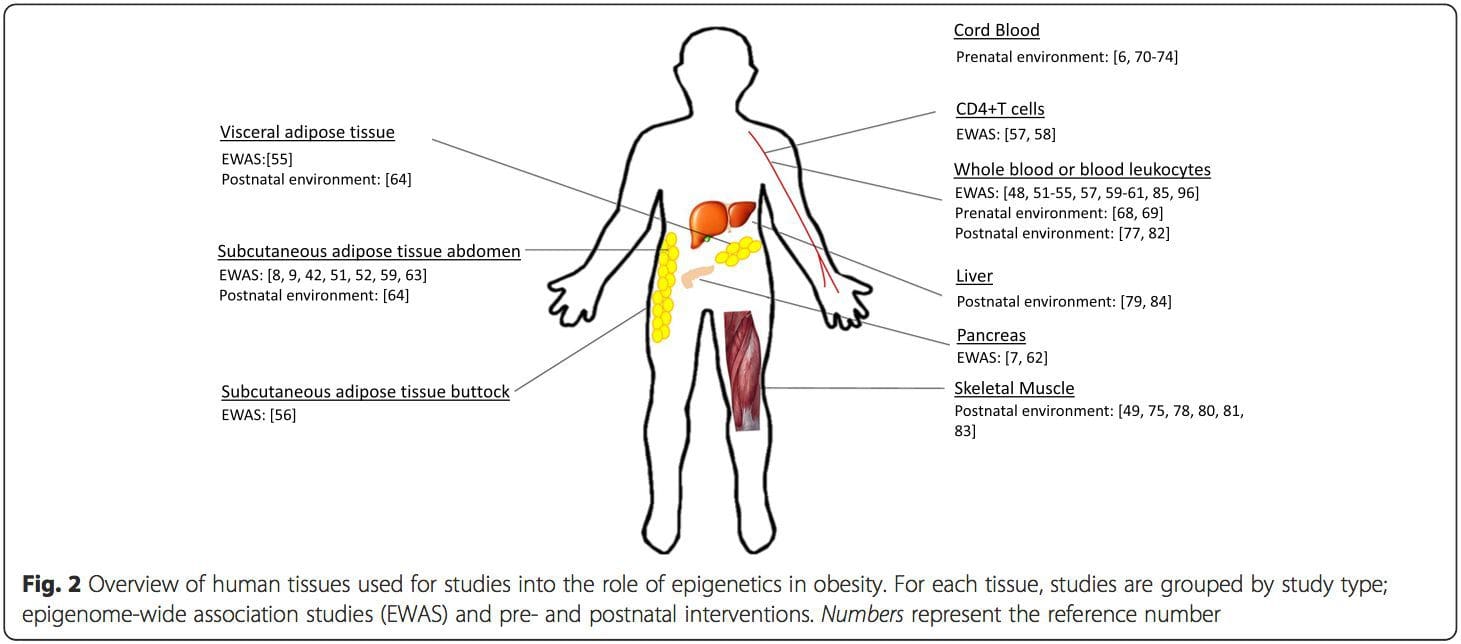 Conclusions
Conclusions





