1 Pirlich M, Schutz T, Norman K, Gastell S,
L�bke HJ, Bischoff SC, Bolder U, Frieling
T, G�ldenzoph H, Hahn K, Jauch KW,
Schindler K, Stein J, Volkert D, Weimann A,
Werner H, Wolf C, Z�rcher G, Bauer P, Lochs
H: The German hospital malnutrition study.
Clin Nutr 2006;25:563�572.
2 Amaral TF, Matos LC, Tavares MM, Subtil
A, Martins R, Nazar� M, Sousa Pereira N:
The economic impact of disease-related malnutrition
at hospital admission. Clin Nutr
2007;26:778�784.
3 Pichard C, Kyle UG, Morabia A, Perrier A,
Vermeulen B, Unger P: Nutritional assessment:
lean body mass depletion at hospital
admission is associated with increased
length of stay. Am J Clin Nutr 2004;79:613�
618.
4 Capuano G, Gentile PC, Bianciardi F, Tosti
M, Palladino A, Di Palma M: Prevalence and
influence of malnutrition on quality of life
and performance status in patients with locally
advanced head and neck cancer before
treatment. Support Care Cancer 2010;18:
433�437.
5 Prado CM, Lieffers JR, McCargar LJ, Reiman
T, Sawyer MB, Martin L, Baracos VE: Prevalence
and clinical implications of sarcopenic
obesity in patients with solid tumours of the
respiratory and gastrointestinal tracts: a
population-based study. Lancet Oncol 2008;
9:629�635.
6 Tan BHL, Birdsell LA, Martin L, Baracos VE,
Fearon KC: Sarcopenia in an overweight or
obese patient is an adverse prognostic factor
in pancreatic cancer. Clin Cancer Res 2009;
15:6973�6979.
7 Baracos VE, Reiman T, Mourtzakis M,
Gioulbasanis I, Antoun S: Body composition
in patients with non-small cell lung cancer:
a contemporary view of cancer cachexia
with the use of computed tomography image
analysis. Am J Clin Nutr 2010;91(suppl):
1133S�1137S.
8 Antoun S, Baracos VE, Birdsell L, Escudier
B, Sawyer MB: Low body mass index and sarcopenia
associated with dose-limiting toxicity
of sorafenib in patients with renal cell carcinoma.
Ann Oncol 2010;21:1594�1598
9 Prado CM, Baracos VE, McCargar LJ,
Mourtzakis M, Mulder KE, Reiman T, Butts
CA, Scarfe AG, Sawyer MB: Body composition
as an independent determinant of 5-fluorouracil-based
chemotherapy toxicity. Clin
Cancer Res 2007;13:3264�3268.
10 Prado CM, Baracos VE, McCargar LJ, Reiman
T, Mourtzakis M, Tonkin K, Mackey JR,
Koski S, Pituskin E, Sawyer MB: Sarcopenia
as a determinant of chemotherapy toxicity
and time to tumor progression in metastatic
breast cancer patients receiving capecitabine
treatment. Clin Cancer Res 2009;15:2920�
2926.
11 Hofhuis JG, Spronk PE, van Stel HF, Schrijvers
GJ, Rommes JH, Bakker J: The impact of
critical illness on perceived health-related
quality of life during ICU treatment, hospital
stay, and after hospital discharge: a longterm
follow-up study. Chest 2008;133:377�
385.
12 Guest JF, Panca M, Baeyens JP, de Man F,
Ljungqvist O, Pichard C, Wait S, Wilson L:
Health economic impact of managing patients
following a community-based diagnosis
of malnutrition in the UK. Clin Nutr 2011;
30:422�429.
13 Kyle UG, Morabia A, Slosman DO, Mensi N,
Unger P, Pichard C: Contribution of body
composition to nutritional assessment at
hospital admission in 995 patients: a controlled
population study. Br J Nutr 2001;86:
725�731.
14 Kondrup J, Allison SP, Elia M; Vellas B,
Plauth M: Educational and Clinical Practice
Committee, European Society of Parenteral
and Enteral Nutrition (ESPEN): ESPEN
guidelines for nutrition screening 2002. Clin
Nutr 2003;22:415�421.
15 Haute Autorit� de Sant�: IPAQSS: informations.
2010. http://www.has-sante.fr/portail/
jcms/c_970427/ipaqss-informations.
16 World Health Organization: Obesity and
overweight: fact sheet No. 311. 2011. http://
www.who.int/mediacentre/factsheets/fs311/
en/index.html.
17 Thibault R, Chikhi M, Clerc A, Darmon P,
Chopard P, Picard-Kossovsky M, Genton L,
Pichard C: Assessment of food intake in hospitalised
patients: a 10 year-comparative
study of a prospective hospital survey. Clin
Nutr 2011;30:289�296.
18 Stenholm S, Harris TB, Rantanen T, Visser
M, Kritchevsky SB, Ferrucci L: Sarcopenic
obesity: definition, cause and consequences.
Curr Opin Clin Nutr Metab Care 2008;11:
693�700.
19 Pichard C, Kyle UG: Body composition measurements
during wasting diseases. Curr
Opin Clin Nutr Metab Care 1998;1:357�361.
20 Wang ZM, Pierson RN Jr, Heymsfield SB:
The five-level model: a new approach to organizing
body-composition research. Am J
Clin Nutr 1992;56:19�28.
21 Schols AM, Broekhuizen R, Weling-Scheepers
CA, Wouters EF: Body composition and
mortality in chronic obstructive pulmonary
disease. Am J Clin Nutr 2005;82:53�59.
22 Slinde F, Gronberg A, Engstrom CP, Rossander-Hulthen
L, Larsson S: Body composition
by bioelectrical impedance predicts
mortality in chronic obstructive pulmonary
disease patients. Respir Med 2005;99:1004�
1009.
23 Vestbo J, Prescott E, Almdal T, Dahl M, Nordestgaard
BG, Andersen T, Sorensen TI,
Lange P: Body mass, fat-free body mass, and
prognosis in patients with chronic obstructive
pulmonary disease from a random population
sample: findings from the Copenhagen
City Heart Study. Am J Respir Crit Care
Med 2006;173:79�83.
24 Segall L, Mardare NG, Ungureanu S, Busuioc
M, Nistor I, Enache R, Marian S, Covic A:
Nutritional status evaluation and survival in
haemodialysis patients in one centre from
Romania. Nephrol Dial Transplant 2009;24:
2536�2540.
25 Beddhu S, Pappas LM, Ramkumar N, Samore
M: Effects of body size and body composition
on survival in hemodialysis patients. J
Am Soc Nephrol 2003;14:2366�2372.
26 F�rstenberg A, Davenport A: Assessment
of body composition in peritoneal dialysis
patients using bioelectrical impedance and
dual-energy X-ray absorptiometry. Am J
Nephrol 2011;33:150�156.
27 Futter JE, Cleland JG, Clark AL: Body mass
indices and outcome in patients with chronic
heart failure. Eur J Heart Fail 2011;13:207�
213.
28 Marin B, Desport JC, Kajeu P, Jesus P, Nicolaud
B, Nicol M, Preux PM, Couratier P: Alteration
of nutritional status at diagnosis is
a prognostic factor for survival of amyotrophic
lateral sclerosis patients. J Neurol
Neurosurg Psychiatry 2011;82:628�634.
29 Janiszewski PM, Oeffinger KC, Church TS,
Dunn AL, Eshelman DA, Victor RG, Brooks
S, Turoff AJ, Sinclair E, Murray JC, Bashore
L, Ross R: Abdominal obesity, liver fat, and
muscle composition in survivors of childhood
acute lymphoblastic leukemia. J Clin
Endocrinol Metab 2007;92:3816�3821.
30 Wagner D, Adunka C, Kniepeiss D, Jakoby
E, Schaffellner S, Kandlbauer M, Fahrleitner-Pammer
A, Roller RE, Kornprat P, M�ller
H, Iberer F, Tscheliessnigg KH: Serum albumin,
subjective global assessment, body
mass index and the bioimpedance analysis in
the assessment of malnutrition in patients up
to 15 years after liver transplantation. Clin
Transplant 2011;25:E396�E400.
31 Kimyagarov S, Klid R, Levenkrohn S, Fleissig
Y, Kopel B, Arad M, Adunsky A: Body
mass index (BMI), body composition and
mortality of nursing home elderly residents.
Arch Gerontol Geriatr 2010;51:227�230.
32 Buffa R, Mereu RM, Putzu PF, Floris G,
Marini E: Bioelectrical impedance vector
analysis detects low body cell mass and dehydration
in patients with Alzheimer�s disease.
J Nutr Health Aging 2010;14:823�827.
33 Schols AM, Wouters EF, Soeters PB, Westerterp
KR: Body composition by bioelectricalimpedance
analysis compared with deuterium
dilution and skinfold anthropometry in
patients with chronic obstructive pulmonary
disease. Am J Clin Nutr 1991;53:421�424.
34 Thibault R, Le Gallic E, Picard-Kossovsky
M, Darmaun D, Chambellan A: Assessment
of nutritional status and body composition
in patients with COPD: comparison of several
methods (in French). Rev Mal Respir
2010;27:693�702.
35 Kyle UG, Janssens JP, Rochat T, Raguso CA,
Pichard C: Body composition in patients
with chronic hypercapnic respiratory failure.
Respir Med 2006;100:244�252.
36 Rieken R, van Goudoever JB, Schierbeek H,
Willemsen SP, Calis EA, Tibboel D, Evenhuis
HM, Penning C: Measuring body composition
and energy expenditure in children with
severe neurologic impairment and intellectual
disability. Am J Clin Nutr 2011;94:759�
766
37 Avram MM, Fein PA, Borawski C, Chattopadhyay
J, Matza B: Extracellular mass/body
cell mass ratio is an independent predictor of
survival in peritoneal dialysis patients. Kidney
Int Suppl 2010;117:S37�S40.
38 Frisancho AR: New norms of upper limb fat
and muscle areas for assessment of nutritional
status. Am J Clin Nutr 1981;34:2540�2545.
39 Caregaro L, Alberino F, Amodio P, Merkel C,
Bolognesi M, Angeli P, Gatta A: Malnutrition
in alcoholic and virus-related cirrhosis.
Am J Clin Nutr l996;63:602�609.
40 Alberino F, Gatta A, Amodio P, Merkel C, Di
Pascoli L, Boffo G, Caregaro L: Nutrition and
survival in patients with liver cirrhosis. Nutrition
2001;17:445�450.
41 Liu E, Spiegelman D, Semu H, Hawkins C,
Chalamilla G, Aveika A, Nyamsangia S,
Mehta S, Mtasiwa D, Fawzi W: Nutritional
status and mortality among HIV-infected
patients receiving antiretroviral therapy in
Tanzania. J Infect Dis 2011;204:282�290.
42 Soler-Cataluna JJ, Sanchez-Sanchez L, Martinez-Garcia
MA, Sanchez PR, Salcedo E,
Navarro M: Mid-arm muscle area is a better
predictor of mortality than body mass index
in COPD. Chest 2005;128:2108�2115.
43 Marquis K, Debigar� R, Lacasse Y, LeBlanc P,
Jobin J, Carrier G, Maltais F: Midthigh muscle
cross-sectional area is a better predictor
of mortality than body mass index in patients
with chronic obstructive pulmonary
disease. Am J Respir Crit Care Med 2002;15;
166:809�813.
44 Kyle UG, Pirlich M, Lochs H, Schuetz T, Pichard
C: Increased length of hospital stay in
underweight and overweight patients at hospital
admission: a controlled population
study. Clin Nutr 2005;24:133�142.
45 Kyle UG, Bosaeus I, De Lorenzo AD, Deurenberg
P, Elia M, G�mez JM, Heitmann
BL, Kent-Smith L, Melchior JC, Pirlich M,
Scharfetter H, Schols AM, Pichard C, Composition
of the ESPEN Working Group. Bioelectrical
impedance analysis. 1. Review of
principles and methods. Clin Nutr 2004;23:
1226�1243.
46 Santarpia L, Marra M, Montagnese C, Alfonsi
L, Pasanisi F, Contaldo F: Prognostic
significance of bioelectrical impedance
phase angle in advanced cancer: preliminary
observations. Nutrition 2009;25:930�931.
47 Gupta D, Lammersfeld CA, Vashi PG, King
J, Dahlk SL, Grutsch JF, Lis CG: Bioelectrical
impedance phase angle in clinical practice:
implications for prognosis in stage IIIB and
IV non-small cell lung cancer. BMC Cancer
2009;9:37.
48 Gupta D, Lis CG, Dahlk SL, Vashi PG,
Grutsch JF, Lammersfeld CA: Bioelectrical
impedance phase angle as a prognostic indicator
in advanced pancreatic cancer. Br J
Nutr 2004;92:957�962.
49 Gupta D, Lammersfeld CA, Burrows JL,
Dahlk SL, Vashi PG, Grutsch JF, Hoffman S,
Lis CG: Bioelectrical impedance phase angle
in clinical practice: implications for prognosis
in advanced colorectal cancer. Am J Clin
Nutr 2004;80:1634�1638.
50 Paiva SI, Borges LR, Halpern-Silveira D, Assun��o
MC, Barros AJ, Gonzalez MC: Standardized
phase angle from bioelectrical impedance
analysis as prognostic factor for
survival in patients with cancer. Support
Care Cancer 2010;19:187�192.
51 Schwenk A, Beisenherz A, R�mer K, Kremer
G, Salzberger B, Elia M: Phase angle from
bioelectrical impedance analysis remains an
independent predictive marker in HIV-infected
patients in the era of highly active antiretroviral
treatment. Am J Clin Nutr 2000;
72:496�501.
52 Desport JC, Marin B, Funalot B, Preux PM,
Couratier P: Phase angle is a prognostic factor
for survival in amyotrophic lateral sclerosis.
Amyotroph Lateral Scler 2008;9:273�
278.
53 Wirth R, Volkert D, R�sler A, Sieber CC,
Bauer JM: Bioelectric impedance phase angle
is associated with hospital mortality of
geriatric patients. Arch Gerontol Geriatr
2010;51:290�294.
54 Mushnick R, Fein PA, Mittman N, Goel N,
Chattopadhyay J, Avram MM: Relationship
of bioelectrical impedance parameters to nutrition
and survival in peritoneal dialysis patients.
Kidney Int Suppl 2003;87:S53�S56.
55 Selberg O, Selberg D: Norms and correlates
of bioimpedance phase angle in healthy human
subjects, hospitalized patients, and patients
with liver cirrhosis. Eur J Appl Physiol
2002;86:509�516.
56 Shah S, Whalen C, Kotler DP, Mayanja H,
Namale A, Melikian G, Mugerwa R, Semba
RD: Severity of human immunodeficiency
virus infection is associated with decreased
phase angle, fat mass and body cell mass in
adults with pulmonary tuberculosis infection
in Uganda. J Nutr 2001;131:2843�2847.
57 Barbosa-Silva MC, Barros AJ: Bioelectric impedance
and individual characteristics as
prognostic factors for post-operative complications.
Clin Nutr 2005;24:830�838.
58 Durnin JV, Womersley J: Body fat assessed
from total body density and its estimation
from skinfold thickness: measurements on
481 men and women aged from 16 to 72
years. Br J Nutr 1974;32:77�97.
59 Hill GL: Body composition research: implications
for the practice of clinical nutrition.
JPEN J Parenter Enter Nutr 1992;16:197�218.
60 Pierson RN Jr, Wang J, Thornton JC, Van
Itallie TB, Colt EW: Body potassium by fourpi
40K counting: an anthropometric correction.
Am J Physiol 1984;246:F234�F239.
61 Sohlstr�m A, Forsum E: Changes in total
body fat during the human reproductive cycle
as assessed by magnetic resonance imaging,
body water dilution, and skinfold thickness:
a comparison of methods. Am J Clin
Nutr 1997;66:1315�1322.
62 Leonard CM, Roza MA, Barr RD, Webber
CE: Reproducibility of DXA measurements
of bone mineral density and body composition
in children. Pediatr Radiol 2009;39:148�
154.
63 Genton L, Karsegard VL, Zawadynski S, Kyle
UG, Pichard C, Golay A, Hans DB: Comparison
of body weight and composition measured
by two different dual energy X-ray absorptiometry
devices and three acquisition
modes in obese women. Clin Nutr 2006;25:
428�437.
64 Jaffrin MY: Body composition determination
by bioimpedance: an update. Curr Opin
Clin Nutr Metab Care 2009;12:482�486.
65 Kyle UG, Pichard C, Rochat T, Slosman DO,
Fitting JW, Thiebaud D: New bioelectrical
impedance formula for patients with respiratory
insufficiency: comparison to dual-energy
X-ray absorptiometry. Eur Respir J 1998;
12:960�966.
66 Kyle UG, Bosaeus I, De Lorenzo AD, Deurenberg
P, Elia M, Manuel G�mez J, Lilienthal
Heitmann B, Kent-Smith L, Melchior
JC, Pirlich M, Scharfetter H, Schols AMWJ,
Pichard C, ESPEN: Bioelectrical impedance
analysis. 2. Utilization in clinical practice.
Clin Nutr 2004;23:1430�1453.
67 Mourtzakis M, Prado CM, Lieffers JR, Reiman
T, McCargar LJ, Baracos VE: A practical
and precise approach to quantification of
body composition in cancer patients using
computed tomography images acquired during
routine care. Appl Physiol Nutr Metab
2008;33:997�1006.
68 Bolton CE, Ionescu AA, Shiels KM, Pettit RJ,
Edwards PH, Stone MD, Nixon LS, Evans
WD, Griffiths TL, Shale DJ: Associated loss
of fat-free mass and bone mineral density in
chronic obstructive pulmonary disease. Am
J Respir Crit Care Med 2004;170:1286�1293.
69 Kyle UG, Genton L, Karsegard L, Slosman
DO, Pichard C: Single prediction equation
for bioelectrical impedance analysis in
adults aged 20�94 years. Nutrition 2001;17:
248�253.
70 Kyle UG, Genton L, Slosman DO, Pichard C:
Fat-free and fat mass percentiles in 5,225
healthy subjects aged 15 to 98 years. Nutrition
2001;17(7�8):534�541.
71 Kotler DP, Burastero S, Wang J, Pierson RN
Jr: Prediction of body cell mass, fat-free
mass, and total body water with bioelectrical
impedance analysis: effects of race, sex, and
disease. Am J Clin Nutr 1996;64:489S�497S.
72 Kyle UG, Genton L, Mentha G, Nicod L, Slosman
DO, Pichard C: Reliable bioelectrical
impedance analysis estimate of fat-free mass
in liver, lung, and heart transplant patients.
JPEN J Parenter Enteral Nutr 2001;25:45�51.
73 Mattar L, Godart N, Melchior JC, Falissard
B, Kolta S, Ringuenet D, Vindreau C, Nordon
C, Blanchet C, Pichard C: Underweight
patients with anorexia nervosa: comparison
of bioelectrical impedance analysis using
five equations to dual X-ray absorptiometry.
Clin Nutr 2011, E-pub ahead of print.
74 Genton L, Karsegard VL, Kyle UG, Hans DB,
Michel JP, Pichard C: Comparison of four
bioelectrical impedance analysis formulas in
healthy elderly subjects. Gerontology 2001;
47:315�323.
75 Robert S, Zarowitz BJ, Hyzy R, Eichenhorn
M, Peterson EL, Popovich J Jr: Bioelectrical
impedance assessment of nutritional status
in critically ill patients. Am J Clin Nutr 1993;
57:840�844.
76 Pichard C, Kyle UG, Slosman DO, Penalosa
B: Energy expenditure in anorexia nervosa:
can fat-free mass as measured by bioelectrical
impedance predict energy expenditure in
hospitalized patients? Clin Nutr 1996;15:
109�114.
77 Kreymann KG, Berger MM, Deutz NE, Hiesmayr
M, Jolliet P, Kazandjiev G, Nitenberg
G, van den Berghe G, Wernerman J, DGEM
(German Society for Nutritional Medicine),
Ebner C, Hartl W, Heymann C, Spies C, ESPEN:
ESPEN guidelines on enteral nutrition:
intensive care. Clin Nutr 2006;25:210�223.
78 Singer P, Berger MM, van den Berghe G, Biolo
G, Calder P, Forbes A, Griffiths R, Kreyman
G, Leverve X, Pichard C, ESPEN: ESPEN
guidelines on parenteral nutrition: intensive
care. Clin Nutr 2009;28:387�400.
79 Magnuson B, Peppard A, Auer Flomenhoft
D: Hypocaloric considerations in patients
with potentially hypometabolic disease
states. Nutr Clin Pract 2011;26:253�260.
80 Rigaud D, Boulier A, Tallonneau I, Brindisi
MC, Rozen R: Body fluid retention and body
weight change in anorexia nervosa patients
during refeeding. Clin Nutr 2010;29:749�
755.
81 Kyle UG, Chalandon Y, Miralbell R, Karsegard
VL, Hans D, Trombetti A, Rizzoli R,
Helg C, Pichard C: Longitudinal follow-up of
body composition in hematopoietic stem cell
transplant patients. Bone Marrow Transplant
2005;35:1171�1177.
82 Pison CM, Cano NJ, Cherion C, Caron F,
Court-Fortune I, Antonini M, GonzalezBermejo
J, Meziane L, Molano LC, Janssens
JP, Costes F, Wuyam B, Similowski T, Melloni
B, Hayot M, Augustin J, Tardif C,
Lejeune H, Roth H, Pichard C, the IRAD Investigators:
Multimodal nutritional rehabilitation
improves clinical outcomes of malnourished
patients with chronic respiratory
failure: a controlled randomised trial. Thorax
2011;66:953�960.
83 Pichard C, Kyle U, Chevrolet JC, Jolliet P,
Slosman D, Mensi N, Temler E, Ricou B: Lack
of effects of recombinant growth hormone
on muscle function in patients requiring
prolonged mechanical ventilation: a prospective,
randomized, controlled study. Crit
Care Med 1996;24:403�413.
84 Pichard C, Kyle UG, Jolliet P, Slosman DO,
Rochat T, Nicod L, Romand J, Mensi N,
Chevrolet JC: Treatment of cachexia with recombinant
growth hormone in a patient before
lung transplantation: a case report. Crit
Care Med 1999;27:1639�1642.
85 Leslie WD, Miller N, Rogala L, Bernstein
CN: Body mass and composition affect bone
density in recently diagnosed inflammatory
bowel disease: the Manitoba IBD Cohort
Study. Inflamm Bowel Dis 2009;15:39�46.
86 van der Meij BS, Langius JA, Smit EF,
Spreeuwenberg MD, von Blomberg BM,
Heijboer AC, Paul MA, van Leeuwen PA:
Oral nutritional supplements containing (n-
3) polyunsaturated fatty acids affect the nutritional
status of patients with stage III nonsmall
cell lung cancer during multimodality
treatment. J Nutr 2010;140:1774�1780.
87 Ryan AM, Reynolds JV, Healy L, Byrne M,
Moore J, Brannelly N, McHugh A, McCormack
D, Flood P: Enteral nutrition enriched
with eicosapentaenoic acid (EPA) preserves
lean body mass following esophageal cancer
surgery: results of a double-blinded randomized
controlled trial. Ann Surg 2009;249:
355�363.
88 Ndekha MJ, Oosterhout JJ, Zijlstra EE, Manary
M, Saloojee H, Manary MJ: Supplementary
feeding with either ready-to-use fortified
spread or corn-soy blend in wasted
adults starting antiretroviral therapy in Malawi:
randomised, investigator blinded, controlled
trial. BMJ 2009;338:b1867�b1875.
89 Ha L, Hauge T, Iversen PO: Body composition
in older acute stroke patients after treatment
with individualized, nutritional supplementation
while in hospital. BMC Geriatrics
2010;10:75.
90 Genton L, Karsegard VL, Chevalley T, Kossovsky
MP, Darmon P, Pichard C: Body
composition changes over 9 years in
healthy elderly subjects and impact of physical
activity. Clin Nutr 2011;30:436�442.
91 Reid CL, Murgatroyd PR, Wright A, Menon
DK: Quantification of lean and fat tissue repletion
following critical illness: a case report.
Crit Care 2008;12:R79.
92 Antoun S, Birdsel Ll, Sawyer MB, Venner P,
Escudier B, Baracos VE: Association of skeletal
muscle wasting with treatment with
sorafenib in patients with advanced renal
cell carcinoma: results from a placebo-controlled
study. J Clin Oncol 2010;28:1054�
1060.
93 Prado CM, Antoun S, Sawyer MB, Baracos
VE: Two faces of drug therapy in cancer:
drug-related lean tissue loss and its adverse
consequences to survival and toxicity. Curr
Opin Clin Nutr Metab Care 2011;14:250�
254.
94 Schutz Y, Kyle UG, Pichard C: Fat-free mass
index and fat mass index percentiles in Caucasians
aged 18�98 y. Int J Obes 2002;26:
953�960.
95 Kyle UG, Schutz Y, Dupertuis YM, Pichard
C: Body composition interpretation: contributions
of the fat-free mass index and the
body fat mass index. Nutrition 2003;19:597�
604.
96 Kyle UG, Piccoli A, Pichard C: Body composition
measurements: interpretation finally
made easy for clinical use. Curr Opin Clin
Nutr Metab Care 2003;6:387�393.


 1. Become An Expert At Chronic Back Pain Management
1. Become An Expert At Chronic Back Pain Management No one pain management technique works for everyone; it helps to be open to trying all sorts of methods and techniques to diminish and manage your pain.
No one pain management technique works for everyone; it helps to be open to trying all sorts of methods and techniques to diminish and manage your pain.


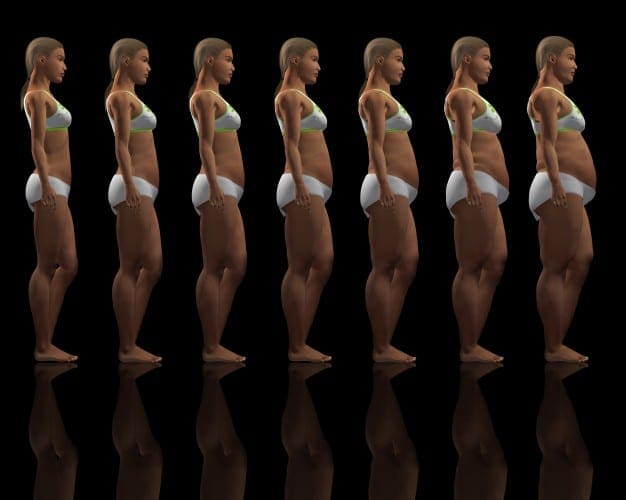
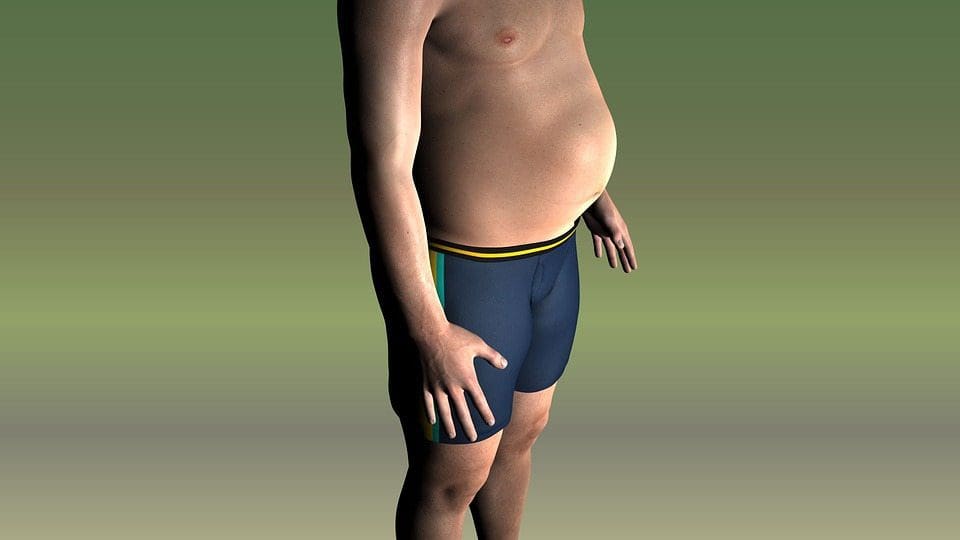 Chronic undernutrition is characterized by a progressive reduction of the�fat-free mass (FFM) and fat mass (FM)�and �which has deleterious consequences on health. Undernutrition is insufficiently screened and treated in hospitalized or at-risk patients despite its high prevalence and negative impact on mortality, morbidity, length of stay (LOS), quality of life, and costs [1�4]. The risk of underestimating hospital undernutrition is likely to worsen in the next decades because of the increasing prevalence of overweight, obesity, and chronic diseases and the increased number of elderly subjects. These clinical conditions are associated with FFM loss (sarcopenia). Therefore, an increased number of patients with FFM loss and sarcopenic obesity will be seen in the future.
Chronic undernutrition is characterized by a progressive reduction of the�fat-free mass (FFM) and fat mass (FM)�and �which has deleterious consequences on health. Undernutrition is insufficiently screened and treated in hospitalized or at-risk patients despite its high prevalence and negative impact on mortality, morbidity, length of stay (LOS), quality of life, and costs [1�4]. The risk of underestimating hospital undernutrition is likely to worsen in the next decades because of the increasing prevalence of overweight, obesity, and chronic diseases and the increased number of elderly subjects. These clinical conditions are associated with FFM loss (sarcopenia). Therefore, an increased number of patients with FFM loss and sarcopenic obesity will be seen in the future.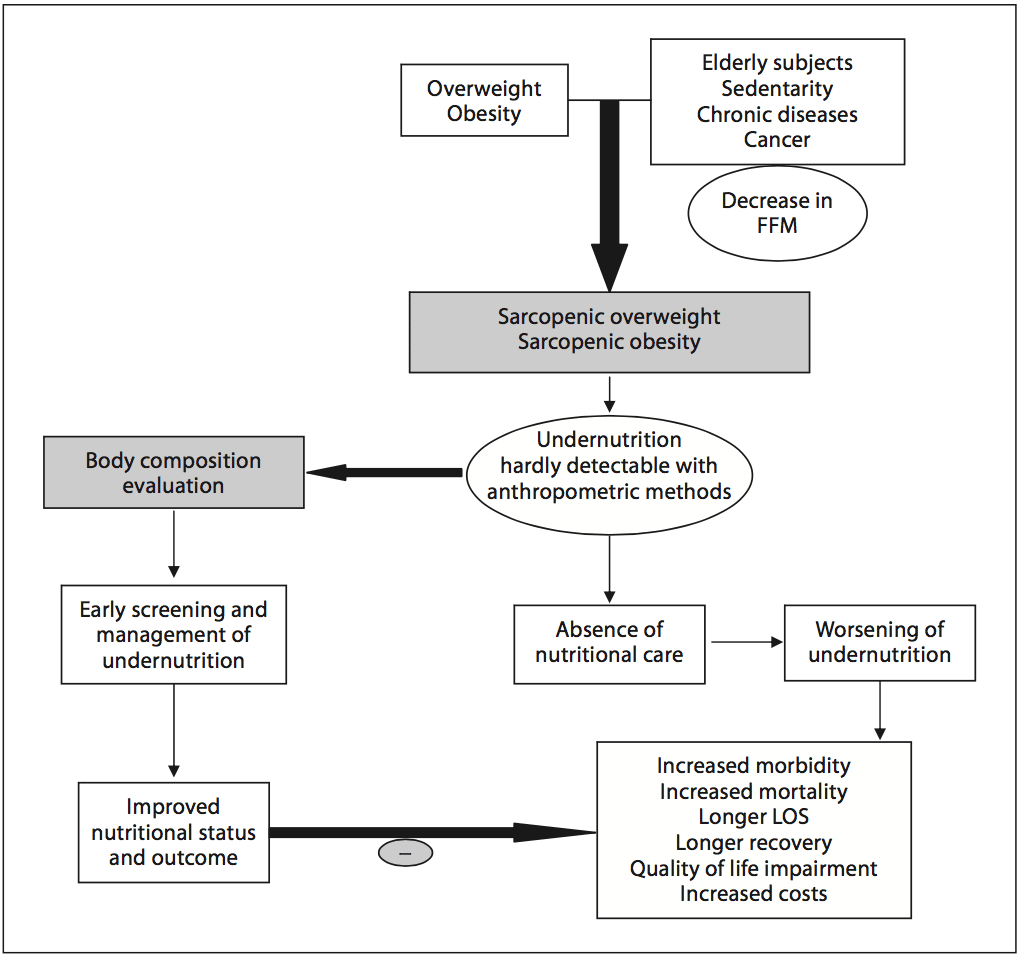
 Academic societies encourage systematic screening of undernutrition at hospital admission and during the hospital stay [14]. The detection of undernutrition is generally based on measurements of weight and height, calculations of BMI, and the percentage of weight loss. Nevertheless, screening of undernutrition is infrequent in hospitalized or nutritionally at-risk ambulatory patients. For example, in France, surveys performed by the French Health Authority [15] indicate that: (i) weight alone, (ii) weight with BMI or percentage of weight loss, and (iii) weight, BMI,�and percentage of weight loss are reported in only 55, 30, and 8% of the hospitalized patients� records, respectively. Several issues, which could be improved by specific educational programs, explain the lack of implementation of nutritional screening in hospitals (table 1). In addition, the accuracy of the clinical screening of undernutrition could be limited at hospital admission. Indeed, patients with undernutrition may have the same BMI as sex- and age- matched healthy controls but a significantly decreased FFM hidden by an expansion of the FM and the total body water which can be measured by bioelectrical impedance analysis (BIA) [13]. This example illustrates that body composition evaluation allows a more accurate identification of FFM loss than body weight loss or BMI decrease. The lack of sensitivity and specificity of weight, BMI, and percentage of weight loss argue for the need for other methods to evaluate the nutritional status.
Academic societies encourage systematic screening of undernutrition at hospital admission and during the hospital stay [14]. The detection of undernutrition is generally based on measurements of weight and height, calculations of BMI, and the percentage of weight loss. Nevertheless, screening of undernutrition is infrequent in hospitalized or nutritionally at-risk ambulatory patients. For example, in France, surveys performed by the French Health Authority [15] indicate that: (i) weight alone, (ii) weight with BMI or percentage of weight loss, and (iii) weight, BMI,�and percentage of weight loss are reported in only 55, 30, and 8% of the hospitalized patients� records, respectively. Several issues, which could be improved by specific educational programs, explain the lack of implementation of nutritional screening in hospitals (table 1). In addition, the accuracy of the clinical screening of undernutrition could be limited at hospital admission. Indeed, patients with undernutrition may have the same BMI as sex- and age- matched healthy controls but a significantly decreased FFM hidden by an expansion of the FM and the total body water which can be measured by bioelectrical impedance analysis (BIA) [13]. This example illustrates that body composition evaluation allows a more accurate identification of FFM loss than body weight loss or BMI decrease. The lack of sensitivity and specificity of weight, BMI, and percentage of weight loss argue for the need for other methods to evaluate the nutritional status. In 2008, twelve and thirty percent of the worldwide adult population was obese or overweight; this is two times higher than in 1980 [16]. The prevalence of overweight and obesity is also increasing in hospitalized patients. A 10-year comparative survey performed in a European hospital showed an increase in patients� BMI, together with a shorter LOS [17]. The BMI increase masks undernutrition and FFM loss at hospital admission. The increased prevalence of obesity in an aging population has led to the recognition of a new nutritional entity: �sarcopenic obesity� [18]. Sarcopenic obesity is characterized by increased FM and reduced FFM with a normal or high body weight. The emergence of the concept of sarcopenic obesity will increase the number of situations associated with a lack of sensitivity of the calculations of BMI and�body weight change for the early detection of FFM loss. This supports a larger use of body composition evaluation for the assessment and follow-up of nutritional status in clinical practice (fig. 1).
In 2008, twelve and thirty percent of the worldwide adult population was obese or overweight; this is two times higher than in 1980 [16]. The prevalence of overweight and obesity is also increasing in hospitalized patients. A 10-year comparative survey performed in a European hospital showed an increase in patients� BMI, together with a shorter LOS [17]. The BMI increase masks undernutrition and FFM loss at hospital admission. The increased prevalence of obesity in an aging population has led to the recognition of a new nutritional entity: �sarcopenic obesity� [18]. Sarcopenic obesity is characterized by increased FM and reduced FFM with a normal or high body weight. The emergence of the concept of sarcopenic obesity will increase the number of situations associated with a lack of sensitivity of the calculations of BMI and�body weight change for the early detection of FFM loss. This supports a larger use of body composition evaluation for the assessment and follow-up of nutritional status in clinical practice (fig. 1).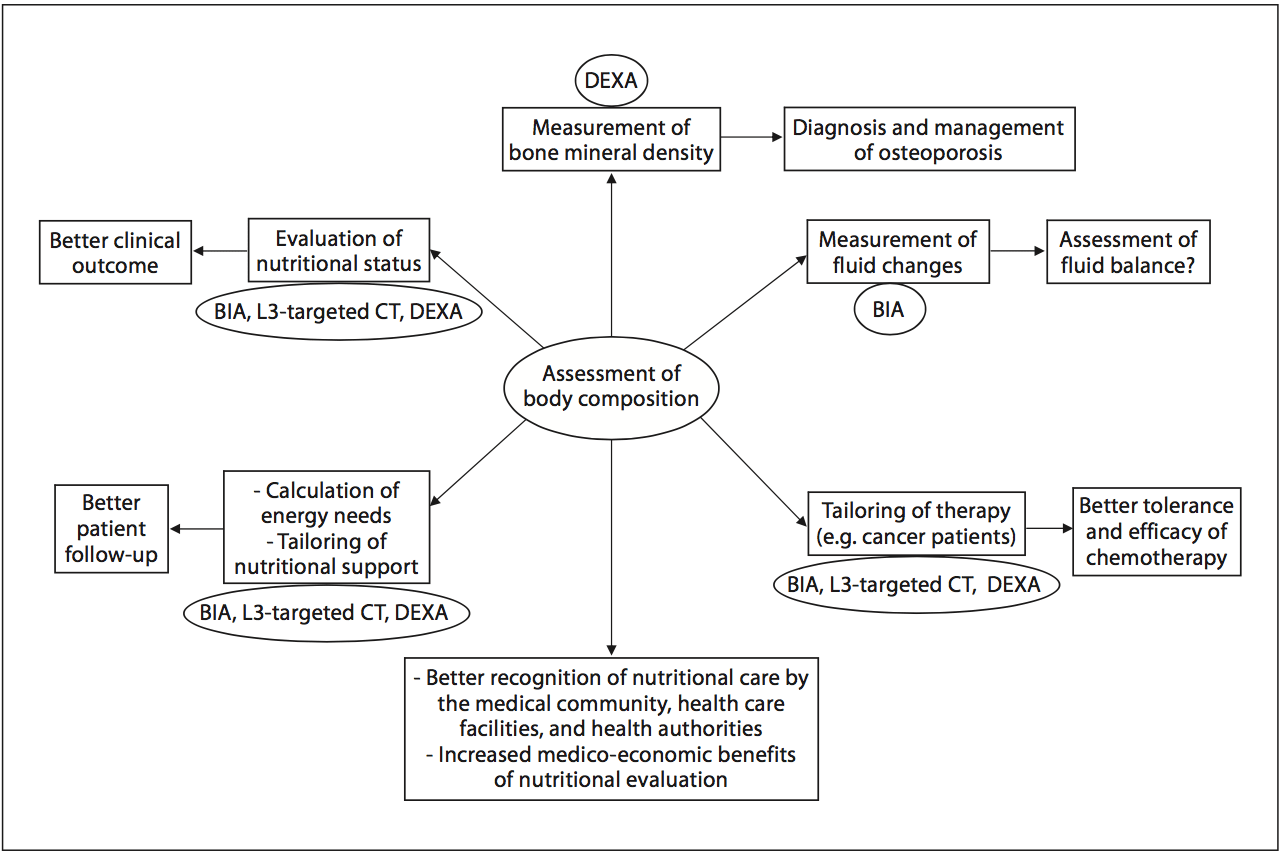 Fig. 2. Current and potential applications of body composition evaluation in clinical practice. The applications are indicated in the boxes, and the body composition methods that could be used for each application are indicated inside the circles. The most used application of body composition evaluation is the measurement of bone mineral density by DEXA for the diagnosis and management of osteoporosis. Although a low FFM is associated with worse clinical outcomes, FFM evaluation is not yet implemented enough in clinical practice. However, by allowing early detection of undernutrition, body composition evaluation could improve the clinical outcome. Body composition evaluation could also be used to follow up nutritional status, calculate energy needs, tailor nutritional support, and assess fluid changes during perioperative period and renal insufficiency. Recent evidence indicates that�a low FFM is associated with a higher toxicity of some chemo- therapy drugs in cancer patients. Thus, by allowing tailoring of the chemotherapy doses to the FFM in cancer patients, body com- position evaluation should improve the tolerance and the efficacy of chemotherapy. BIA, L3-targeted CT, and DEXA could be used for the assessment of nutritional status, the calculation of energy needs, and the tailoring of nutritional support and therapy. Further studies are warranted to validate BIA as an accurate method for fluid balance measurement. By integrating body composition evaluation into the management of different clinical conditions, all of these potential applications would lead to a better recognition of nutritional care by the medical community, the health care facilities, and the health authorities, as well as to an increase in the medico-economic benefits of the nutritional evaluation.
Fig. 2. Current and potential applications of body composition evaluation in clinical practice. The applications are indicated in the boxes, and the body composition methods that could be used for each application are indicated inside the circles. The most used application of body composition evaluation is the measurement of bone mineral density by DEXA for the diagnosis and management of osteoporosis. Although a low FFM is associated with worse clinical outcomes, FFM evaluation is not yet implemented enough in clinical practice. However, by allowing early detection of undernutrition, body composition evaluation could improve the clinical outcome. Body composition evaluation could also be used to follow up nutritional status, calculate energy needs, tailor nutritional support, and assess fluid changes during perioperative period and renal insufficiency. Recent evidence indicates that�a low FFM is associated with a higher toxicity of some chemo- therapy drugs in cancer patients. Thus, by allowing tailoring of the chemotherapy doses to the FFM in cancer patients, body com- position evaluation should improve the tolerance and the efficacy of chemotherapy. BIA, L3-targeted CT, and DEXA could be used for the assessment of nutritional status, the calculation of energy needs, and the tailoring of nutritional support and therapy. Further studies are warranted to validate BIA as an accurate method for fluid balance measurement. By integrating body composition evaluation into the management of different clinical conditions, all of these potential applications would lead to a better recognition of nutritional care by the medical community, the health care facilities, and the health authorities, as well as to an increase in the medico-economic benefits of the nutritional evaluation.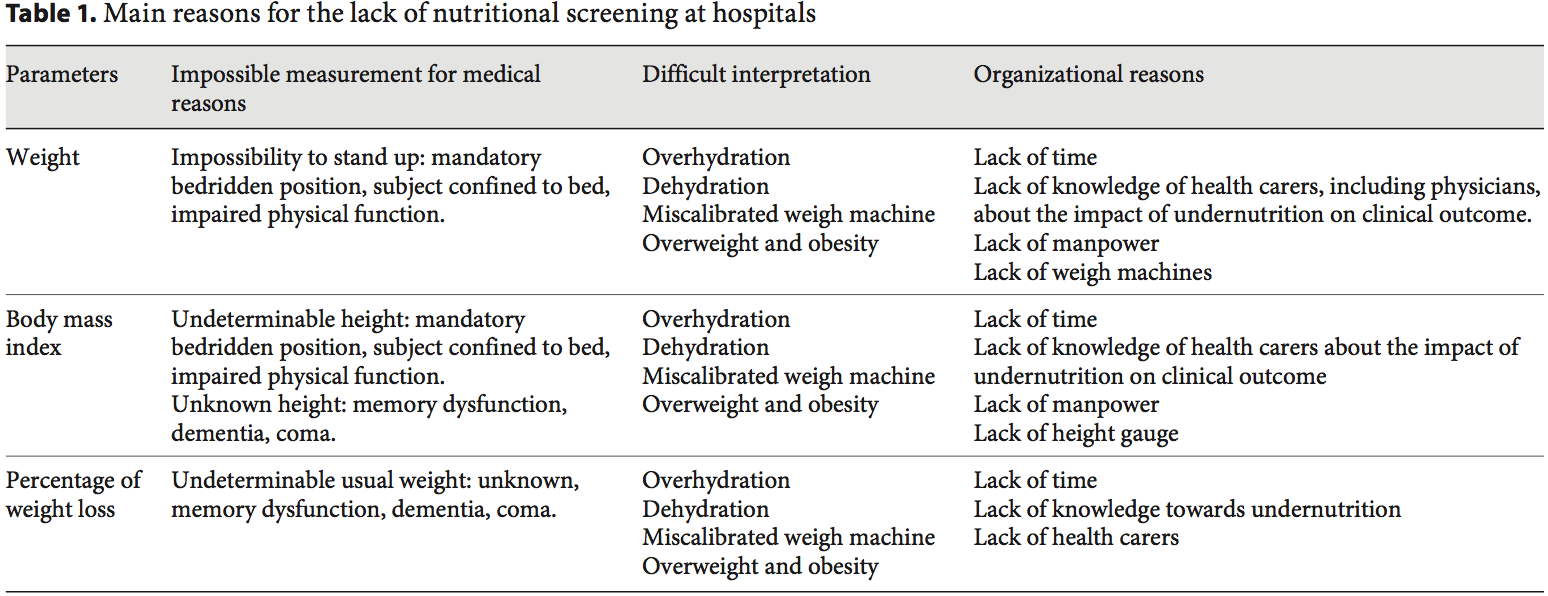
 Body Composition Techniques For FFM Measurement
Body Composition Techniques For FFM Measurement Body composition evaluation allows measurement of the major body compartments: FFM (including bone mineral tissue), FM, and total body water. Table 2 shows indicative values of the body composition of a healthy subject weighing 70 kg. In several clinical situations, i.e. hospital admission, chronic obstructive pulmonary dis- ease (COPD) [21�23], dialysis [24�26], chronic heart failure [27], amyotrophic lateral sclerosis [28], cancer [5, 29], liver transplantation [30], nursing home residence [31], and Alzheimer�s disease [32], changes in body compartments are detected with the techniques of body composition evaluation. At hospital admission, body composition evaluation could be used for the detection of FFM loss and undernutrition. Indeed, FFM and the FFM index (FFMI) [FFM (kg)/height (m2)] measured by BIA are significantly lower in hospitalized patients (n = 995) than in age-, height-, and sex-matched controls (n = 995) [3]. Conversely, clinical tools of nutritional status assessment, such as BMI, subjective global assessment, or mini-nutritional assessment, are not accurate enough to estimate FFM loss and nutritional status [30, 32�34]. In 441 patients with non-small cell lung cancer, FFM loss deter- mined by computerized tomography (CT) was observed in each BMI category [7], and in young adults with all�types of cancer, an increase in FM together with a de- crease in FFM were reported [29]. These findings reveal the lack of sensitivity of BMI to detect FFM loss. More- over, the FFMI is a more sensitive determinant of LOS than a weight loss over 10% or a BMI below 20 [3]. In COPD, the assessment of FFM by BIA is a more sensitive method to detect undernutrition than anthropometry [33, 35]. BIA is also more accurate at assessing nutrition- al status in children with severe neurologic impairment than the measurement of skin fold thickness [36].
Body composition evaluation allows measurement of the major body compartments: FFM (including bone mineral tissue), FM, and total body water. Table 2 shows indicative values of the body composition of a healthy subject weighing 70 kg. In several clinical situations, i.e. hospital admission, chronic obstructive pulmonary dis- ease (COPD) [21�23], dialysis [24�26], chronic heart failure [27], amyotrophic lateral sclerosis [28], cancer [5, 29], liver transplantation [30], nursing home residence [31], and Alzheimer�s disease [32], changes in body compartments are detected with the techniques of body composition evaluation. At hospital admission, body composition evaluation could be used for the detection of FFM loss and undernutrition. Indeed, FFM and the FFM index (FFMI) [FFM (kg)/height (m2)] measured by BIA are significantly lower in hospitalized patients (n = 995) than in age-, height-, and sex-matched controls (n = 995) [3]. Conversely, clinical tools of nutritional status assessment, such as BMI, subjective global assessment, or mini-nutritional assessment, are not accurate enough to estimate FFM loss and nutritional status [30, 32�34]. In 441 patients with non-small cell lung cancer, FFM loss deter- mined by computerized tomography (CT) was observed in each BMI category [7], and in young adults with all�types of cancer, an increase in FM together with a de- crease in FFM were reported [29]. These findings reveal the lack of sensitivity of BMI to detect FFM loss. More- over, the FFMI is a more sensitive determinant of LOS than a weight loss over 10% or a BMI below 20 [3]. In COPD, the assessment of FFM by BIA is a more sensitive method to detect undernutrition than anthropometry [33, 35]. BIA is also more accurate at assessing nutrition- al status in children with severe neurologic impairment than the measurement of skin fold thickness [36]. FFM loss is correlated with survival in different clinical settings [5, 21�28, 37]. In patients with amyotrophic lateral sclerosis, an FM increase, but not an FFM in- crease, measured by BIA, was correlated with survival during the course of the disease [28]. The relation between body composition and mortality has not yet been demonstrated in the intensive care unit. The relation between body composition and mortality has been demonstrated with anthropometric methods, BIA, and CT. Measurement of the mid-arm muscle circumference is an easy tool to diagnose sarcopenia [38]. The mid-arm muscle circumference has been shown to be correlated with survival in patients with cirrhosis [39, 40], HIV infection [41], and COPD in a stronger way than BMI [42]. The relation between FFM loss and mortality has been extensively shown with BIA [21�28, 31, 37], which is the most used method. Recently, very interesting data suggest that CT could evaluate the disease prognosis in relation to muscle wasting. In obese cancer patients, sarcopenia as assessed by CT measurement of the total skeletal muscle cross-sectional area is an independent predictor of the survival of patients with bronchopulmonary [5, 7], gastrointestinal [5], and pancreatic cancers [6]. FFM assessed by measurement of the mid-thigh muscle cross- sectional area by CT is also predictive of mortality in COPD patients with severe chronic respiratory insufficiency [43]. In addition to mortality, a low FFMI at hospital admission is significantly associated with an in- creased LOS [3, 44]. A bicentric controlled population study performed in 1,717 hospitalized patients indicates that both loss of FFM and excess of FM negatively affect the LOS [44]. Patients with sarcopenic obesity are most at risk of increased LOS. This study also found that ex- cess FM reduces the sensitivity of BMI to detect nutritional depletion [44]. Together with the observation that the BMI of hospitalized patients has increased during the last decade [17], these findings suggest that FFM and�FFMI measurement should be used to evaluate nutritional status in hospitalized patients.
FFM loss is correlated with survival in different clinical settings [5, 21�28, 37]. In patients with amyotrophic lateral sclerosis, an FM increase, but not an FFM in- crease, measured by BIA, was correlated with survival during the course of the disease [28]. The relation between body composition and mortality has not yet been demonstrated in the intensive care unit. The relation between body composition and mortality has been demonstrated with anthropometric methods, BIA, and CT. Measurement of the mid-arm muscle circumference is an easy tool to diagnose sarcopenia [38]. The mid-arm muscle circumference has been shown to be correlated with survival in patients with cirrhosis [39, 40], HIV infection [41], and COPD in a stronger way than BMI [42]. The relation between FFM loss and mortality has been extensively shown with BIA [21�28, 31, 37], which is the most used method. Recently, very interesting data suggest that CT could evaluate the disease prognosis in relation to muscle wasting. In obese cancer patients, sarcopenia as assessed by CT measurement of the total skeletal muscle cross-sectional area is an independent predictor of the survival of patients with bronchopulmonary [5, 7], gastrointestinal [5], and pancreatic cancers [6]. FFM assessed by measurement of the mid-thigh muscle cross- sectional area by CT is also predictive of mortality in COPD patients with severe chronic respiratory insufficiency [43]. In addition to mortality, a low FFMI at hospital admission is significantly associated with an in- creased LOS [3, 44]. A bicentric controlled population study performed in 1,717 hospitalized patients indicates that both loss of FFM and excess of FM negatively affect the LOS [44]. Patients with sarcopenic obesity are most at risk of increased LOS. This study also found that ex- cess FM reduces the sensitivity of BMI to detect nutritional depletion [44]. Together with the observation that the BMI of hospitalized patients has increased during the last decade [17], these findings suggest that FFM and�FFMI measurement should be used to evaluate nutritional status in hospitalized patients. Numerous methods of body composition evaluation have been developed: anthropometry, including the 4-skinfold method [58], hydrodensitometry [58], in vivo neutron activation analysis [59], anthropogammametry from total body potassium-40 [60], nuclear magnetic resonance [61], dual-energy X-ray absorptiometry (DEXA) [62, 63], BIA [45, 64�66], and more recently CT [7, 43, 67]. DEXA, BIA, and CT appear to be the most convenient methods for clinical practice (fig. 2), while the other methods are reserved for scientific use.
Numerous methods of body composition evaluation have been developed: anthropometry, including the 4-skinfold method [58], hydrodensitometry [58], in vivo neutron activation analysis [59], anthropogammametry from total body potassium-40 [60], nuclear magnetic resonance [61], dual-energy X-ray absorptiometry (DEXA) [62, 63], BIA [45, 64�66], and more recently CT [7, 43, 67]. DEXA, BIA, and CT appear to be the most convenient methods for clinical practice (fig. 2), while the other methods are reserved for scientific use. The evaluation of FFM could be used for the calculation of energy needs, thus allowing the optimization of nutritional intakes according to nutritional needs. This could be of great interest in specific situations, such as severe neurologic disability, overweight, and obesity. In 61 children with severe neurologic impairment and intellectual disability, an equation integrating body composition had good agreement with the doubly labeled water method. It gave a better estimation of energy expenditure than did the Schofield predictive equation [36]. However, in 9 anorexia nervosa patients with a mean BMI of 13.7, pre- diction formulas of resting energy expenditure including FFM did not allow accurate prediction of the resting energy expenditure measured by indirect calorimetry [76]. In overweight or obese patients, the muscle catabolism in response to inflammation was the same as that observed�in patients with normal BMI. Indeed, despite a higher BMI, the FFM of overweight or obese individuals is similar (or slightly increased) to that of patients with normal BMI. Thus, the use of actual weight for the assessment of the energy needs of obese patients would result in over- feeding and its related complications. Therefore, the ex- perts recommend the use of indirect calorimetry or calculation of the energy needs of overweight or obese patients as follows: 15 kcal/kg actual weight/day or 20�25 kcal/kg ideal weight/day [77, 78], although these predictive formulas could be inaccurate in some clinical conditions [79]. In a US prospective study conducted in 33 ICU medical and surgical ventilated ICU patients, daily measurement of the active cell mass (table 2) by BIA was used to assess the adequacy between energy/protein intakes and needs. In that study, nutritional support with 30 kcal/ kg actual body weight/day energy and 1.5 g/kg/day protein allowed stabilization of the active cell mass [75]. Thus, follow-up of FFM by BIA could help optimize nutritional intakes when indirect calorimetry cannot be performed.
The evaluation of FFM could be used for the calculation of energy needs, thus allowing the optimization of nutritional intakes according to nutritional needs. This could be of great interest in specific situations, such as severe neurologic disability, overweight, and obesity. In 61 children with severe neurologic impairment and intellectual disability, an equation integrating body composition had good agreement with the doubly labeled water method. It gave a better estimation of energy expenditure than did the Schofield predictive equation [36]. However, in 9 anorexia nervosa patients with a mean BMI of 13.7, pre- diction formulas of resting energy expenditure including FFM did not allow accurate prediction of the resting energy expenditure measured by indirect calorimetry [76]. In overweight or obese patients, the muscle catabolism in response to inflammation was the same as that observed�in patients with normal BMI. Indeed, despite a higher BMI, the FFM of overweight or obese individuals is similar (or slightly increased) to that of patients with normal BMI. Thus, the use of actual weight for the assessment of the energy needs of obese patients would result in over- feeding and its related complications. Therefore, the ex- perts recommend the use of indirect calorimetry or calculation of the energy needs of overweight or obese patients as follows: 15 kcal/kg actual weight/day or 20�25 kcal/kg ideal weight/day [77, 78], although these predictive formulas could be inaccurate in some clinical conditions [79]. In a US prospective study conducted in 33 ICU medical and surgical ventilated ICU patients, daily measurement of the active cell mass (table 2) by BIA was used to assess the adequacy between energy/protein intakes and needs. In that study, nutritional support with 30 kcal/ kg actual body weight/day energy and 1.5 g/kg/day protein allowed stabilization of the active cell mass [75]. Thus, follow-up of FFM by BIA could help optimize nutritional intakes when indirect calorimetry cannot be performed. Body composition evaluation allows a qualitative assessment of body weight variations. The evaluation of body composition may help to document the efficiency of nutritional support during a patient�s follow-up of numerous clinical conditions, such as surgery [59], anorexia nervosa [76, 80], hematopoietic stem cell transplantation [81], COPD [82], ICU [83], lung transplantation [84], ulcerative colitis [59], Crohn�s disease [85], cancer [86, 87], HIV/AIDS [88], and acute stroke in elderly patients [89]. Body composition evaluation could be used for the follow-up of healthy elderly subjects [90]. Body composition evaluation allows characterization of the increase in body mass in terms of FFM and FM [81, 91]. After hematopoietic stem cell transplantation, the increase in BMI is the result of the increase in FM, but not of the increase in FFM [81]. Also, during recovery after an acute illness, weight gain 6 months after ICU discharge could be mostly related to an increase in FM (+7 kg) while FFM only increased by 2 kg; DEXA and air displacement plethysmography were used to measure the FM and FFM [91]. These two examples suggest that body composition evaluation could be helpful to decide the modification and/or the renewal of nutritional support. By identifying the patients gaining weight but reporting no or insufficient FFM, body composition evaluation could contribute to influencing the medical decision of continuing nutrition- al support that would have been stopped in the absence of body composition evaluation.
Body composition evaluation allows a qualitative assessment of body weight variations. The evaluation of body composition may help to document the efficiency of nutritional support during a patient�s follow-up of numerous clinical conditions, such as surgery [59], anorexia nervosa [76, 80], hematopoietic stem cell transplantation [81], COPD [82], ICU [83], lung transplantation [84], ulcerative colitis [59], Crohn�s disease [85], cancer [86, 87], HIV/AIDS [88], and acute stroke in elderly patients [89]. Body composition evaluation could be used for the follow-up of healthy elderly subjects [90]. Body composition evaluation allows characterization of the increase in body mass in terms of FFM and FM [81, 91]. After hematopoietic stem cell transplantation, the increase in BMI is the result of the increase in FM, but not of the increase in FFM [81]. Also, during recovery after an acute illness, weight gain 6 months after ICU discharge could be mostly related to an increase in FM (+7 kg) while FFM only increased by 2 kg; DEXA and air displacement plethysmography were used to measure the FM and FFM [91]. These two examples suggest that body composition evaluation could be helpful to decide the modification and/or the renewal of nutritional support. By identifying the patients gaining weight but reporting no or insufficient FFM, body composition evaluation could contribute to influencing the medical decision of continuing nutrition- al support that would have been stopped in the absence of body composition evaluation. In clinical situations when weight and BMI do not reflect the FFM, the evaluation of body composition should be used to adapt drug doses to the FFM and/or FM absolute values in every patient. This point has been recently illustrated in oncology patients with sarcopenic obesity. FFM loss was determined by CT as described above. In cancer patients, some therapies could affect body com- position by inducing muscle wasting [92]. In patients with advanced renal cell carcinoma [92], sorafenib induces a significant 8% loss of skeletal muscular mass at 12 months. In turn, muscle wasting in patients with BMI less than 25 was significantly associated with sorafenib toxicity in patients with metastatic renal cancer [8]. In metastatic breast cancer patients receiving capecitabine treatment, and in patients with colorectal cancer receiving 5-fluorouracile, using the convention of dosing per unit of body surface area, FFM loss was the determinant of chemotherapy toxicity [9, 10] and time to tumor progression [10]. In colorectal cancer patients administered 5-fluoruracil, low FFM is a significant predictor of toxicity only in female patients [9]. The variation in toxicity between women and men may be partially explained by the fact that FFM was lower in females. Indeed, FFM rep- resents the distribution volume of most cytotoxic chemo- therapy drugs. In 2,115 cancer patients, the individual variations in FFM could change by up to three times the distribution volume of the chemotherapy drug per body area unit [5]. Thus, administering the same doses of chemotherapy drugs to a patient with a low FFM compared to a patient with a normal FFM would increase the risk of chemotherapy toxicity [5]. These data suggest that FFM loss could have a direct impact on the clinical outcome of cancer patients. Decreasing chemotherapy doses in case of FFM loss could contribute to improving cancer patients� prognosis through the improvement of the tolerance of chemotherapy. These findings justify the systematic evaluation of body composition in all cancer patients in order to detect FFM loss, tailor chemotherapy doses according to FFM values, and then improve the efficacy- tolerance and cost-efficiency ratios of the therapeutic strategies [93]. Body composition evaluation should also be used to tailor the doses of drugs which are calculated based on patients� weight, e.g. corticosteroids, immuno-suppressors (infliximab, azathioprine or methotrexate), or sedatives (propofol).
In clinical situations when weight and BMI do not reflect the FFM, the evaluation of body composition should be used to adapt drug doses to the FFM and/or FM absolute values in every patient. This point has been recently illustrated in oncology patients with sarcopenic obesity. FFM loss was determined by CT as described above. In cancer patients, some therapies could affect body com- position by inducing muscle wasting [92]. In patients with advanced renal cell carcinoma [92], sorafenib induces a significant 8% loss of skeletal muscular mass at 12 months. In turn, muscle wasting in patients with BMI less than 25 was significantly associated with sorafenib toxicity in patients with metastatic renal cancer [8]. In metastatic breast cancer patients receiving capecitabine treatment, and in patients with colorectal cancer receiving 5-fluorouracile, using the convention of dosing per unit of body surface area, FFM loss was the determinant of chemotherapy toxicity [9, 10] and time to tumor progression [10]. In colorectal cancer patients administered 5-fluoruracil, low FFM is a significant predictor of toxicity only in female patients [9]. The variation in toxicity between women and men may be partially explained by the fact that FFM was lower in females. Indeed, FFM rep- resents the distribution volume of most cytotoxic chemo- therapy drugs. In 2,115 cancer patients, the individual variations in FFM could change by up to three times the distribution volume of the chemotherapy drug per body area unit [5]. Thus, administering the same doses of chemotherapy drugs to a patient with a low FFM compared to a patient with a normal FFM would increase the risk of chemotherapy toxicity [5]. These data suggest that FFM loss could have a direct impact on the clinical outcome of cancer patients. Decreasing chemotherapy doses in case of FFM loss could contribute to improving cancer patients� prognosis through the improvement of the tolerance of chemotherapy. These findings justify the systematic evaluation of body composition in all cancer patients in order to detect FFM loss, tailor chemotherapy doses according to FFM values, and then improve the efficacy- tolerance and cost-efficiency ratios of the therapeutic strategies [93]. Body composition evaluation should also be used to tailor the doses of drugs which are calculated based on patients� weight, e.g. corticosteroids, immuno-suppressors (infliximab, azathioprine or methotrexate), or sedatives (propofol).
 The implementation of body composition evaluation in routine care presents a challenge for the next decades. Indeed the concomitant increases in elderly subjects and patients with chronic diseases and cancer, and in the prevalence of overweight and obesity in the population, will increase the number of patients nutritionally at risk or undernourished, particularly those with sarcopenic obesity. Body composition evaluation should be used to improve the screening of undernutrition in hospitalized patients. The results of body composition should be based on the same principle as BMI calculation, towards the systematic normalization for body height of FFM (FFMI) and FM [FM (kg)/height (m)2 = FM index] [94]. The results could be expressed according to previously de- scribed percentiles of healthy subjects [95, 96]. Body com- position evaluation should be performed at the different stages of the disease, during the course of treatments and the rehabilitation phase. Such repeated evaluations of body composition could allow assessment of the nutritional status, adjusting the calculation of energy needs as kilocalories/kilogram FFM, following the efficacy of
The implementation of body composition evaluation in routine care presents a challenge for the next decades. Indeed the concomitant increases in elderly subjects and patients with chronic diseases and cancer, and in the prevalence of overweight and obesity in the population, will increase the number of patients nutritionally at risk or undernourished, particularly those with sarcopenic obesity. Body composition evaluation should be used to improve the screening of undernutrition in hospitalized patients. The results of body composition should be based on the same principle as BMI calculation, towards the systematic normalization for body height of FFM (FFMI) and FM [FM (kg)/height (m)2 = FM index] [94]. The results could be expressed according to previously de- scribed percentiles of healthy subjects [95, 96]. Body com- position evaluation should be performed at the different stages of the disease, during the course of treatments and the rehabilitation phase. Such repeated evaluations of body composition could allow assessment of the nutritional status, adjusting the calculation of energy needs as kilocalories/kilogram FFM, following the efficacy of 


 Anabolic-Androgenic Steroids
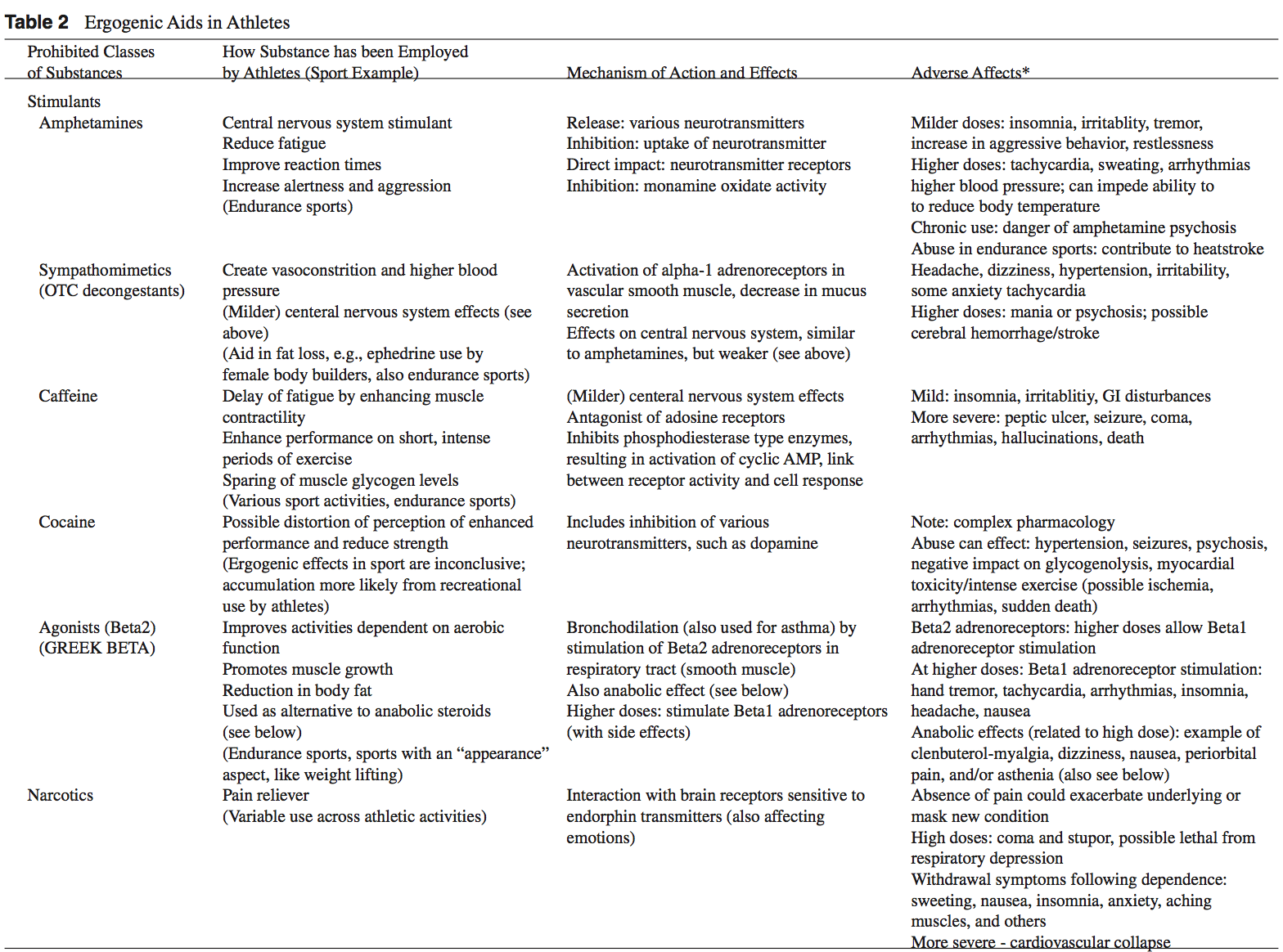
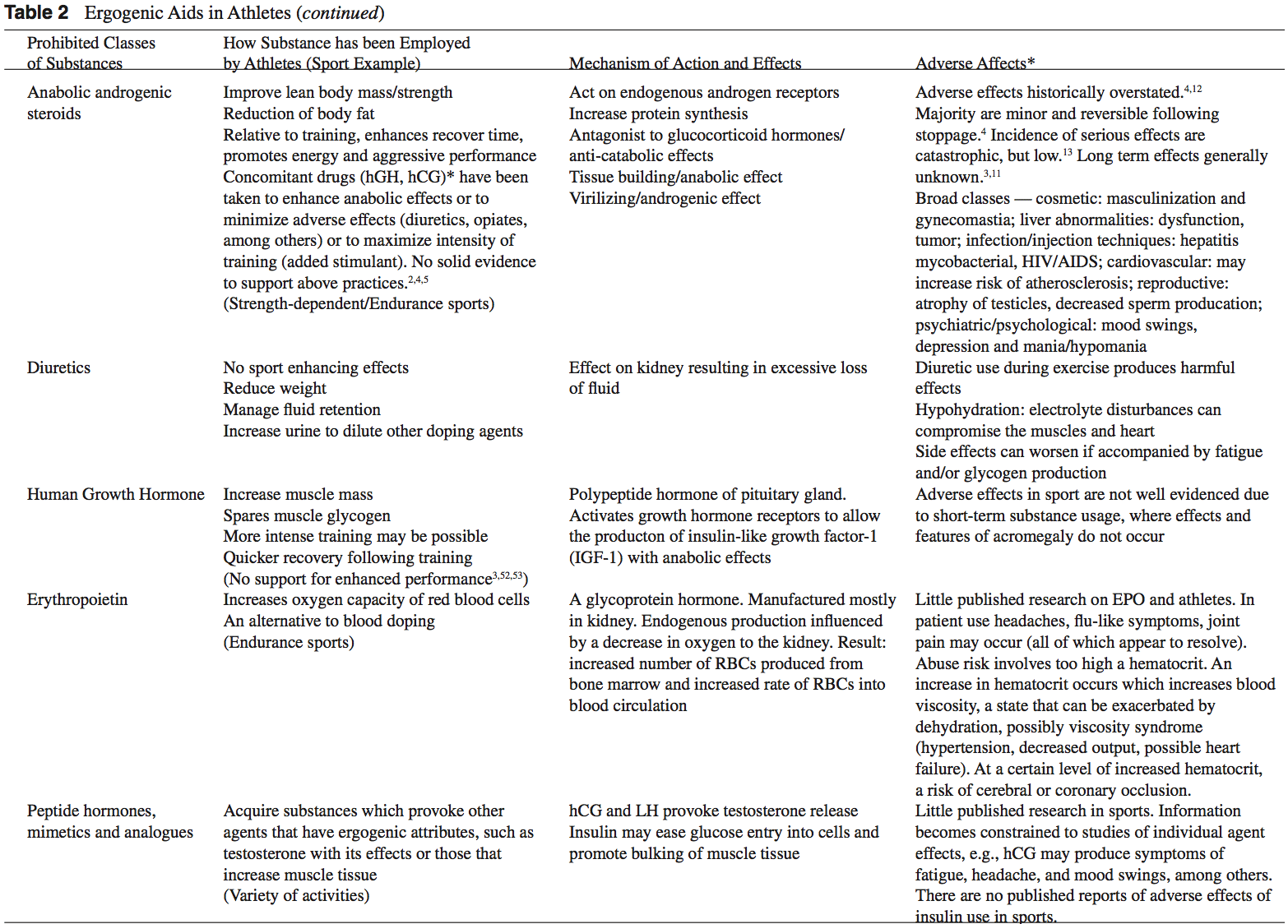
Anabolic-Androgenic Steroids Anabolic-androgenic steroids (AAS) are testosterone derivatives that exert anabolic (tissue building) and androgenic (masculinizing) influences on the body.3 Since the discovery of the chemical structure of testosterone in 1935, attempts to separate the anabolic and androgenic effects of AAS�have been unsuccessful.3
Anabolic-androgenic steroids (AAS) are testosterone derivatives that exert anabolic (tissue building) and androgenic (masculinizing) influences on the body.3 Since the discovery of the chemical structure of testosterone in 1935, attempts to separate the anabolic and androgenic effects of AAS�have been unsuccessful.3  Dehydroepiandrosterone (DHEA) is a precursor to testos- terone produced primarily in the adrenal glands.4,14 Natural sources of DHEA include wild yams. The FDA banned sale of DHEA in 1996 due to insuf cient evidence of safety and value; however, DHEA remains a legal and popular item sold as a nutritional supplement.14,15
Dehydroepiandrosterone (DHEA) is a precursor to testos- terone produced primarily in the adrenal glands.4,14 Natural sources of DHEA include wild yams. The FDA banned sale of DHEA in 1996 due to insuf cient evidence of safety and value; however, DHEA remains a legal and popular item sold as a nutritional supplement.14,15 Androstenedione is a testosterone pre-cursor produced in the adrenal glands and gonads. Several professional athletes have used this substance, bringing it to national attention.2 Androstenedione is found naturally in the pollen of Scottish pine trees.19
Androstenedione is a testosterone pre-cursor produced in the adrenal glands and gonads. Several professional athletes have used this substance, bringing it to national attention.2 Androstenedione is found naturally in the pollen of Scottish pine trees.19 The increased visibility of ergogenic aids in the last de- cade has occurred primarily because of the passage of the United States Dietary Supplement Health and Education Act (DSHEA) of 1994.22 Certain vitamins, minerals, amino acids, herbs, and other botanical preparations can be classified as a �dietary supplement� under the DSHEA guidelines. Dietary supplements, as a result of DSHEA, are no longer under the direct regulatory control of the FDA. In fact, substances sold as a dietary supplement do not require FDA evaluation for safety or efficacy, and do not have to meet quality control standards expected of approved drugs.5 The content and purity of dietary supplements are not regulated and can vary widely.5,23 Since androstenedione and DHEA have been found to occur naturally in plant sources, these testosterone precursors can be labeled as �dietary supplements� and sold legally over-the-counter.
The increased visibility of ergogenic aids in the last de- cade has occurred primarily because of the passage of the United States Dietary Supplement Health and Education Act (DSHEA) of 1994.22 Certain vitamins, minerals, amino acids, herbs, and other botanical preparations can be classified as a �dietary supplement� under the DSHEA guidelines. Dietary supplements, as a result of DSHEA, are no longer under the direct regulatory control of the FDA. In fact, substances sold as a dietary supplement do not require FDA evaluation for safety or efficacy, and do not have to meet quality control standards expected of approved drugs.5 The content and purity of dietary supplements are not regulated and can vary widely.5,23 Since androstenedione and DHEA have been found to occur naturally in plant sources, these testosterone precursors can be labeled as �dietary supplements� and sold legally over-the-counter. Dietary supplements containing Chinese ephedra, also known as Mahaung, are marketed as performance enhancers and weight-loss aids.24 Ephedra species of herb have been used for over 5,000 years for respiratory ailments.25 Currently, ephedrine alkaloids are found in hundreds of prescriptions and over-the-counter products, such as antihistamines, decongestants, and appetite suppressants.24-26 Ephedra and related ephedrine alkaloids are sympathomimetic agents that�mimic epinephrine effects.
Dietary supplements containing Chinese ephedra, also known as Mahaung, are marketed as performance enhancers and weight-loss aids.24 Ephedra species of herb have been used for over 5,000 years for respiratory ailments.25 Currently, ephedrine alkaloids are found in hundreds of prescriptions and over-the-counter products, such as antihistamines, decongestants, and appetite suppressants.24-26 Ephedra and related ephedrine alkaloids are sympathomimetic agents that�mimic epinephrine effects. Creatine use in athletes has grown as a result of a 1992 study that showed that creatine supplementation produced a 20% increase in skeletal muscle creatine concentration.2,35 In the phosphorylated form, creatine serves as an energy substrate that contributes to adenosine triphosphate (ATP) re-synthesis during high-intensity exercise.36 Creatine re- mains popular with power and resistance athletes as it is thought to produce increases in strength, muscle mass, and to delay fatigue.2,14,36
Creatine use in athletes has grown as a result of a 1992 study that showed that creatine supplementation produced a 20% increase in skeletal muscle creatine concentration.2,35 In the phosphorylated form, creatine serves as an energy substrate that contributes to adenosine triphosphate (ATP) re-synthesis during high-intensity exercise.36 Creatine re- mains popular with power and resistance athletes as it is thought to produce increases in strength, muscle mass, and to delay fatigue.2,14,36 Human growth hormone (hGH) is a polypeptide produced in the anterior pituitary gland. After its release from the pituitary, hGH can exert its effect in all cells of the body via tissue specific receptors. Human growth hormone is shown to promote protein anabolism, carbohydrate tolerance, lipolysis, natriuresis, and bone and connective tissue turnover.4,50
Human growth hormone (hGH) is a polypeptide produced in the anterior pituitary gland. After its release from the pituitary, hGH can exert its effect in all cells of the body via tissue specific receptors. Human growth hormone is shown to promote protein anabolism, carbohydrate tolerance, lipolysis, natriuresis, and bone and connective tissue turnover.4,50 Recombinant EPO (r-EPO) was approved by the FDA for manufacture in 1989 after the EPO gene was cloned in 1985.14 Since its approval, r-EPO has been abused for athletic personal gain as an alternative to blood doping.3,14 Recombinant EPO has largely replaced the practice of blood doping, as r-EPO produces a dose-dependent increase in hematocrit.2 In theory, r-EPO should provide all of the benefits of blood doping without the risks involved in blood transfusion.3
Recombinant EPO (r-EPO) was approved by the FDA for manufacture in 1989 after the EPO gene was cloned in 1985.14 Since its approval, r-EPO has been abused for athletic personal gain as an alternative to blood doping.3,14 Recombinant EPO has largely replaced the practice of blood doping, as r-EPO produces a dose-dependent increase in hematocrit.2 In theory, r-EPO should provide all of the benefits of blood doping without the risks involved in blood transfusion.3 The antioxidant capabilities of certain vitamins are believed by many to counter-act the production of free-radials that occurs during exercise.14 Most of the research to date involves vitamin E, vitamin C, and beta carotene.2 The existing literature does not support the notion that antioxidants have significant ergogenic capabilities.2,14,57 There are currently no recommendations for antioxidant use in athletes that exceeds the normal adult recommended daily allowance (RDA).
The antioxidant capabilities of certain vitamins are believed by many to counter-act the production of free-radials that occurs during exercise.14 Most of the research to date involves vitamin E, vitamin C, and beta carotene.2 The existing literature does not support the notion that antioxidants have significant ergogenic capabilities.2,14,57 There are currently no recommendations for antioxidant use in athletes that exceeds the normal adult recommended daily allowance (RDA). Beta-hydroxy-beta-methylbutyrate (HMB) is a metabolite of the branched-chain amino acid leucine. HMB is theorized to inhibit muscle breakdown during strenuous exercise but its exact mechanism of action remains unknown.14,58 Studies show that HMB supplementation may significantly lower serum lactate dehydrogenase (LDH), lower serum creatine phosphokinase (CPK) levels and delay blood lactate accumulation after endurance training compared to placebo.59,60 Furthermore, short-term HMB use has been shown to significantly increase strength gains with resistance-exercised training over placebo in one double-blinded study.61
Beta-hydroxy-beta-methylbutyrate (HMB) is a metabolite of the branched-chain amino acid leucine. HMB is theorized to inhibit muscle breakdown during strenuous exercise but its exact mechanism of action remains unknown.14,58 Studies show that HMB supplementation may significantly lower serum lactate dehydrogenase (LDH), lower serum creatine phosphokinase (CPK) levels and delay blood lactate accumulation after endurance training compared to placebo.59,60 Furthermore, short-term HMB use has been shown to significantly increase strength gains with resistance-exercised training over placebo in one double-blinded study.61 Caffeine is a methylxanthine occurring naturally in many species of plants. Caffeine is thought to work through a variety of mechanisms. The central nervous system effect of caffeine is probably the result of adrenergic receptor antagonism.3 Its use by athletes stems from the theory that caffeine may delay fatigue by enhancing skeletal muscle contractility and spare muscle glycogen levels by enhancing fat metabolism.6 Multiple studies have reported an improved endurance time with caffeine use.6,62,63 There is evidence that caffeine use may enhance performance with more intense short-duration exercise as well.2 The caffeine dosages most associated with an ergogenic effect range in the literature from 3 to 9 mg per kilogram of body weight.2,6
Caffeine is a methylxanthine occurring naturally in many species of plants. Caffeine is thought to work through a variety of mechanisms. The central nervous system effect of caffeine is probably the result of adrenergic receptor antagonism.3 Its use by athletes stems from the theory that caffeine may delay fatigue by enhancing skeletal muscle contractility and spare muscle glycogen levels by enhancing fat metabolism.6 Multiple studies have reported an improved endurance time with caffeine use.6,62,63 There is evidence that caffeine use may enhance performance with more intense short-duration exercise as well.2 The caffeine dosages most associated with an ergogenic effect range in the literature from 3 to 9 mg per kilogram of body weight.2,6








 DD Palmer wrote a chapter in 1914 entitled Inflammation, stating that, “inflammation is present in most, if not all diseases, in the acute if not chronic.” 100 years later, all chronic diseases are inflammatory conditions within local tissues . Chronic systemic inflammation and related pathophysiological changes involve structures and functions that are silent.
DD Palmer wrote a chapter in 1914 entitled Inflammation, stating that, “inflammation is present in most, if not all diseases, in the acute if not chronic.” 100 years later, all chronic diseases are inflammatory conditions within local tissues . Chronic systemic inflammation and related pathophysiological changes involve structures and functions that are silent. Loss of sleep, sedentary living, stress and diet all encourage inflammation.
Loss of sleep, sedentary living, stress and diet all encourage inflammation. Laboratory tests are the best approach to spot chronic inflammation. High levels of glucose, triglycerides, hemoglobin A1c protein along with reduced levels of vitamin D and HDL cholesterol are the most reliable in identifying chronic inflammation.
Laboratory tests are the best approach to spot chronic inflammation. High levels of glucose, triglycerides, hemoglobin A1c protein along with reduced levels of vitamin D and HDL cholesterol are the most reliable in identifying chronic inflammation. Diet is a problem for most people with over two-thirds of the population being overweight. Obesity is known to be a chronic inflammatory state associated with chronic pain and degenerative diseases.
Diet is a problem for most people with over two-thirds of the population being overweight. Obesity is known to be a chronic inflammatory state associated with chronic pain and degenerative diseases.