Assessment and Treatment of Scalenes
These assessment and treatment recommendations represent a synthesis of information derived from personal clinical experience and from the numerous sources which are cited, or are based on the work of researchers, clinicians and therapists who are named (Basmajian 1974, Cailliet 1962, Dvorak & Dvorak 1984, Fryette 1954, Greenman 1989, 1996, Janda 1983, Lewit 1992, 1999, Mennell 1964, Rolf 1977, Williams 1965).
Clinical Application of Neuromuscular Techniques: Scalenes
Box 4.9 Notes on Scalenes
- The scalenes are a controversial muscle since they seem to be both postural and phasic (Lin et al 1994), their status being modified by the type(s) of stress to which they are exposed (see Ch. 3 for discussion of this topic).
- Janda (1988) reports that �spasm and/or trigger points are commonly present in the scalenes as also are weakness and/or inhibition�.
- The attachment sites of the scalene muscles vary, as does their presence. The scalene posterior is sometimes absent, and sometimes blends with the fibres of medius.
- Scalene medius is noted to frequently attach to the atlas (Gray 1995) and sometimes extend to the 2nd rib (Simons et al 1998).
- The scalene minimus (pleuralis), which attaches to the pleural dome, is present in onethird (Platzer 1992) to three-quarters (Simons et al 1998) of people, on at least one side and, when absent, is replaced by a transverse cupular ligament (Platzer 1992).
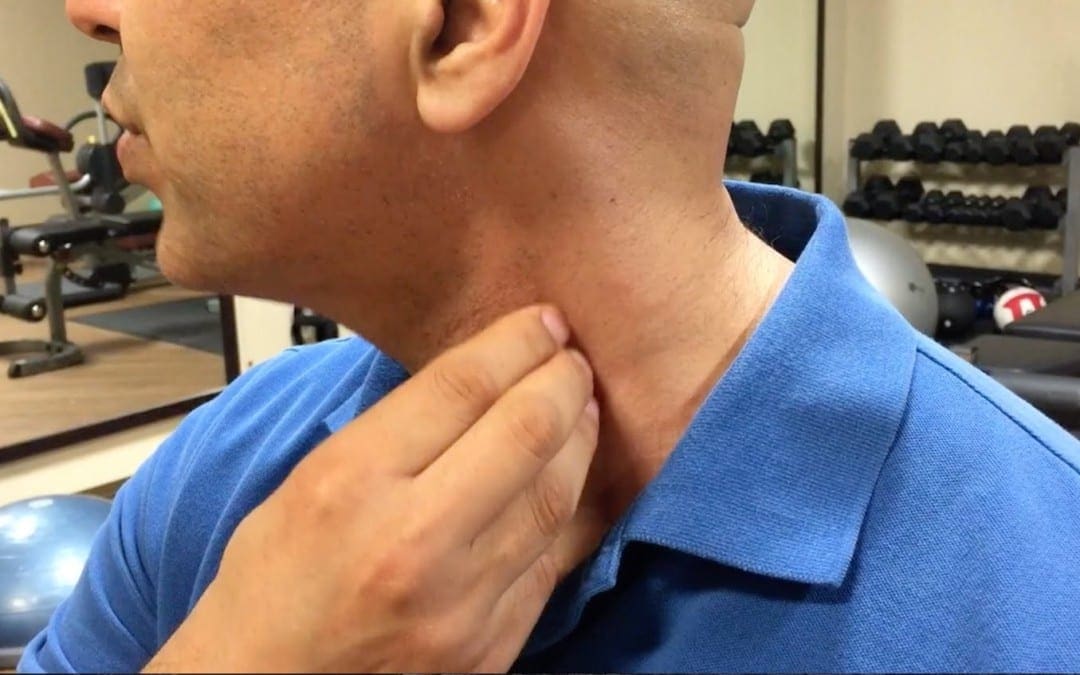
- The brachial plexus exits the cervical column between the scalenus anterior and medius. These two muscles, together with the 1st rib, form the scalene hiatus (also called the �scalene opening� or �posterior scalene aperture�) (Platzer 1992). It is through this opening�that the brachial plexus and vascular structures for the upper extremity pass. When scalene fibres are taut, they may entrap the nerves (scalene anticus syndrome) or may elevate the 1st rib against the clavicle and indirectly crowd the vascular, or neurologic, structures (simultaneous compromising of both neural and vascular structures is rare) (Stedman 1998). Any of these conditions may be diagnosed as �thoracic outlet syndrome�, which is �a collective title for a number of conditions attributed to compromise of blood vessels or nerve fibers (brachial plexus) at any point between the base of the neck and the axilla� (Stedman 1998).
Assessment of Shortness in Scalenes (14)
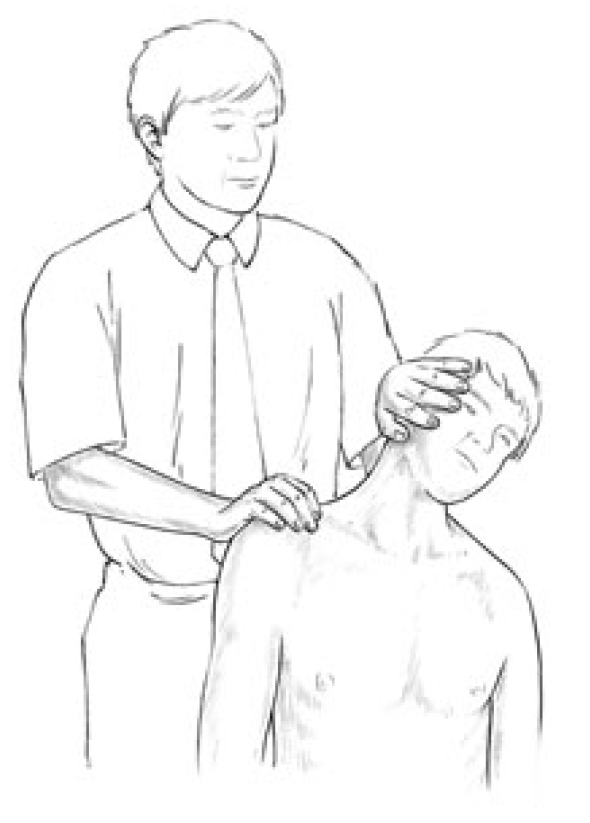
Assessment of cervical sidebending (lateral flexion) strength. This involves the scalenes and levator scapulae (and to a secondary degree the rectus capitis lateralis and the transversospinalis group).
The practitioner places a stabilising hand on the top of the shoulder to prevent movement and the other on the head above the ear, as the seated patient attempts to flex the head laterally against this resistance. Both sides are assessed.
Observation assessment (a) There is no easy test for shortness of the scalenes apart from observation, palpation and assessment of trigger point activity/tautness and a functional observation as follows:
- In most people who have marked scalene shortness there is a tendency to overuse these (and other upper fixators of the shoulder and neck) as accessory breathing muscles.
- There may also be a tendency to hyperventilation (and hence for there to possibly be a history of anxiety, phobic behaviour, panic attacks and/or fatigue symptoms).
- These muscles seem to be excessively tense in many people with chronic fatigue symptoms.
The observation assessment consists of the practitioner placing his relaxed hands over the patient�s shoulders so that the fingertips rest on the clav-icles, at which time the seated patient is asked to inhale deeply. If the practitioner�s hands noticeably rise towards the patient�s ears during inhalation then there exists inappropriate use of scalenes, which indicates that they are stressed, which also means that, by definition, they will have become shortened and require stretching treatment.
Observation assessment (b) (Fig. 4.33) Alternatively, during the history taking interview, the patient can be asked to place one hand on the abdomen just above the umbilicus and the other flat against the upper chest.
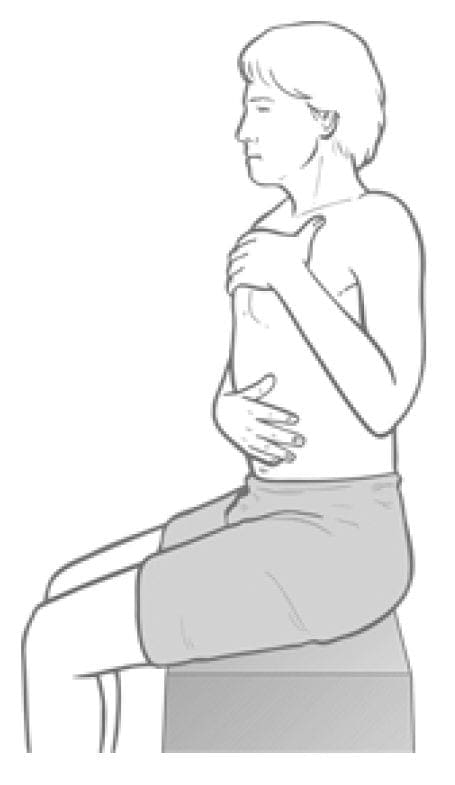
Figure 4.33 Observation assessment of respiratory function. Any tendency for the upper hand to move cephalad, or earlier than the caudad hand, suggests scalene overactivity.
On inhalation, the hands are observed: if the upper hand initiates the breathing process and rises significantly towards the chin, rather than moving forwards, a pattern of upper chest breathing can be assumed, and therefore stress, and therefore shortness of the scalenes (and other accessory breathing muscles, notably sternomastoid).
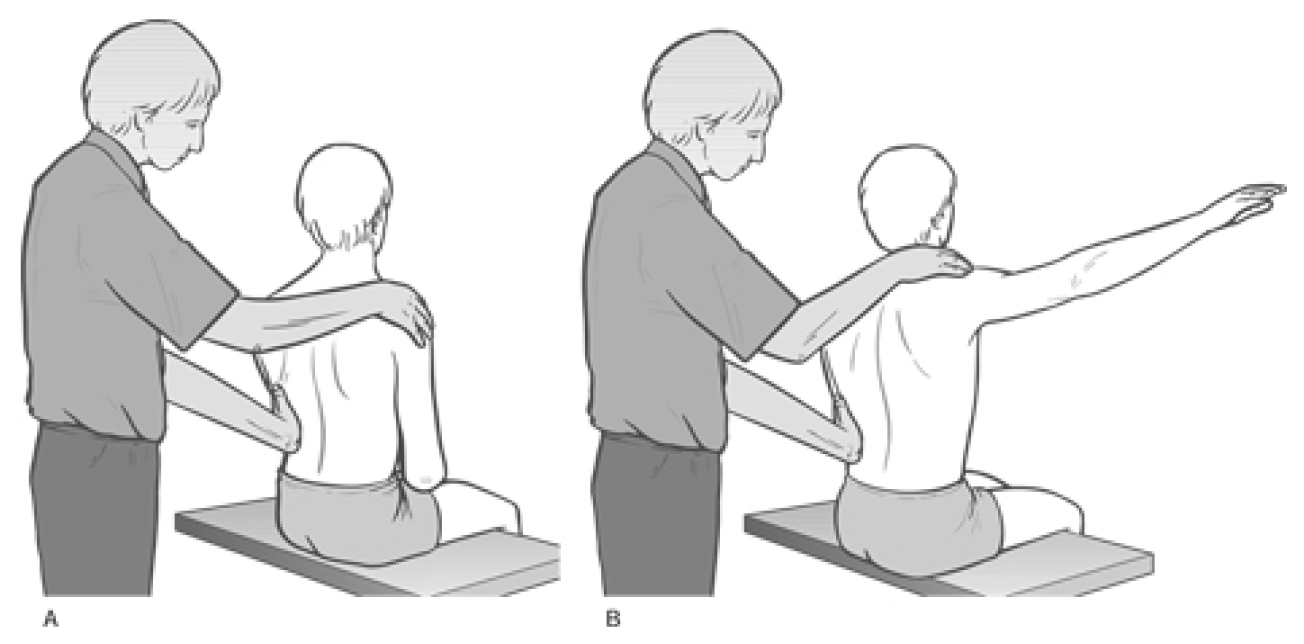
MET Treatment of Short Scalenes (Fig. 4.34A, B, C)
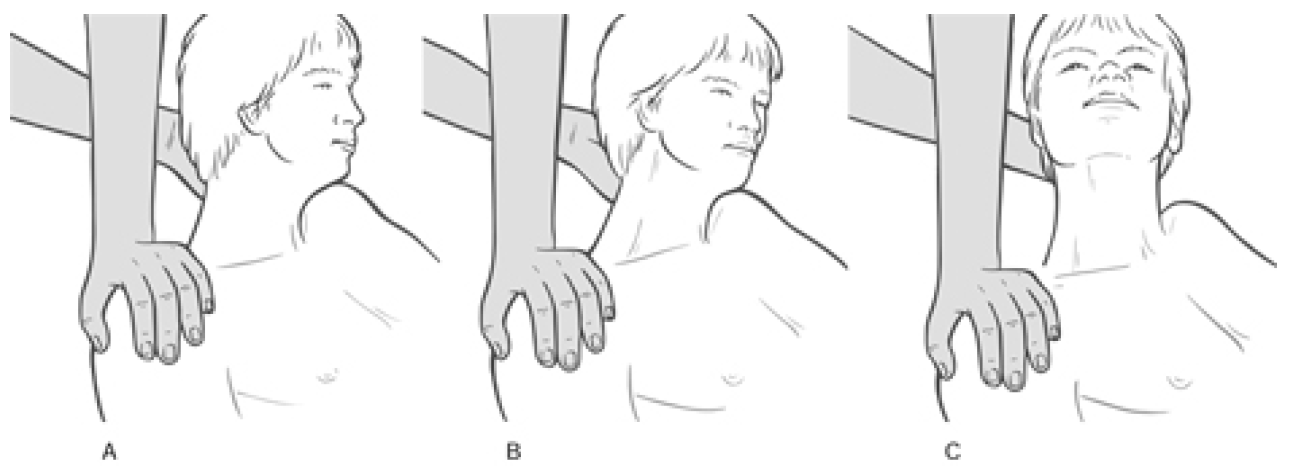
Patient lies supine with a cushion or folded towel under the upper thoracic area so that, unless supported by the practitioner�s contralateral hand, the head would fall into extension. The head is rotated contralaterally (away from the side to be treated). There are three positions of rotation required:
- Full contralateral rotation of the head/neck produces involvement of the more posterior fibres of the scalenes
- A contralateral 45� rotation of the head/neck involves the middle fibres
- A position of only slight contralateral rotation involves the more anterior fibres.
The practitioner�s free hand is placed on the side of the patient�s head to restrain the isometric contraction which will be used to release the scalenes. The patient�s head is in one of the above degrees of rotation, supported by the practitioner�s contralateral hand.
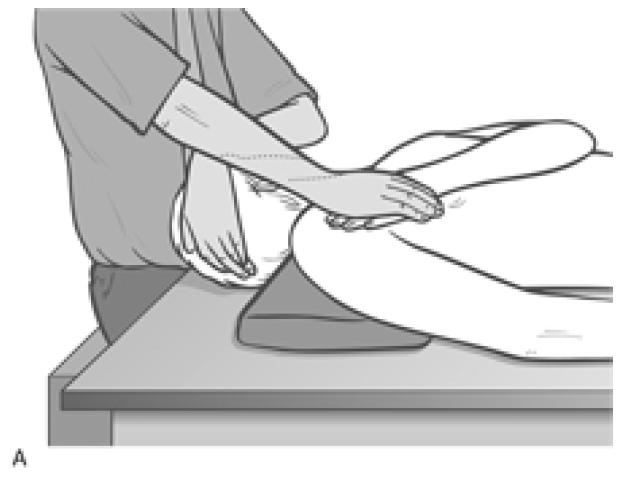
Figure 4.34A MET for scalenus posticus. On stretching, following the isometric contraction, the neck is allowed to move into slight extension while a mild stretch is introduced by the contact hand which rests on the second rib, below the lateral aspect of the clavicle.
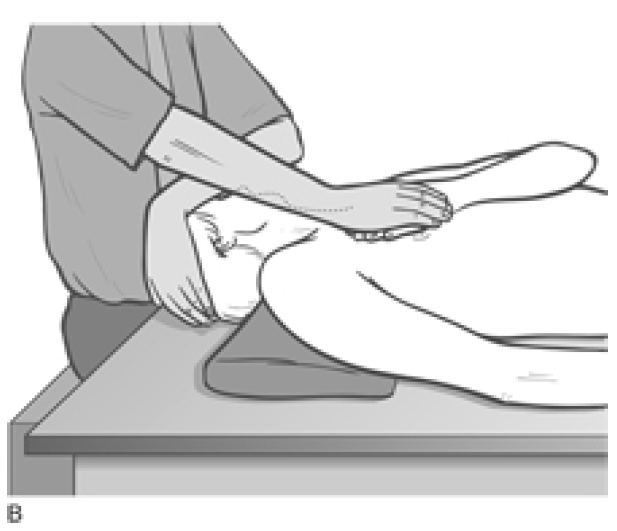
Figure 4.34B MET treatment for the middle fibres of scalenes. The hand placement (thenar or hypothenar eminence of relaxed hand) is on the 2nd rib below the centre of the clavicle.
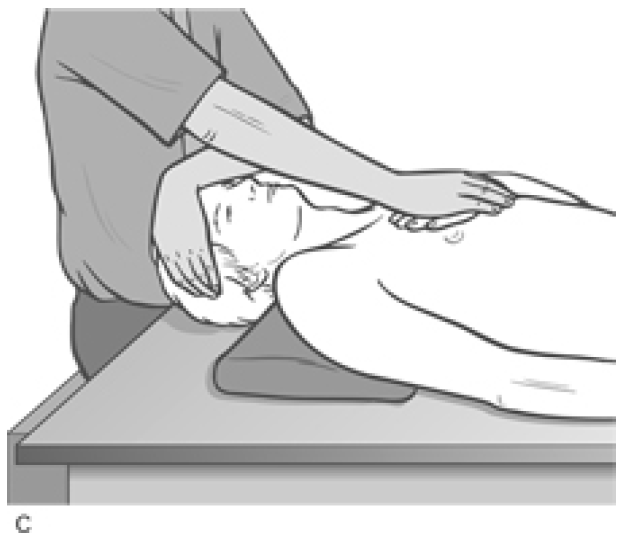
Figure 4.34C MET treatment of the anterior fibres of the scalenes; hand placement is on the sternum
The patient is instructed to try to lift the forehead a fraction and to attempt to turn the head towards the affected side, with appropriate breathing cooperation, while resistance is applied by the practitioner�s hand to prevent both movements (�breathe in and hold your breath as you �lift and turn�, and hold this for 7�10 seconds�). Both the effort and, the counter-pressure should be modest and painless at all times.
After a 7�10 second contraction, the head is placed into extension and one hand remains on it to prevent movement during the scalene stretch.
The patient�s contralateral hand is placed (palm down) just inferior to the lateral end of the clavicle on the affected side (for full rotation of the head, posterior scalenes). The practitioner�s hand which was acting to produce resistance to the isometric contraction is now placed onto the dorsum of the patient�s �cushion� hand.
As the patient slowly exhales, the practitioner�s contact hand, resting on the patient�s hand, which is itself resting on the 2nd rib and upper thorax, pushes obliquely away and towards the foot on that same side, following the rib movement into its exhalation position, so stretching the attached musculature and fascia. This stretch is held for at least 20 seconds after each isometric contraction. The process is then repeated at least once more.
The head is rotated 45� contralaterally and the �cushion� hand contact, which applies the stretch of the middle scalenes, is placed just inferior to the middle aspect of the clavicle. When the head is in the almost upright facing position for the anterior scalene stretch, the �cushion� hand contact is on the upper sternum itself.
In all other ways the methodology is as described for the first position above.
NOTE: It is important not to allow heroic degrees of neck extension during any phase of this treatment. There should be some extension, but it should be appropriate to the age and condition of the individual.
A degree of eye movement can assist scalene treatment and may be used as an alternative to the �lift and turn� muscular effort described above. If the patient makes the eyes look caudally (towards the feet) and towards the affected side during the isometric contraction, she will increase the degree of contraction in the muscles. If during the resting phase, when stretch is being introduced, she looks away from the treated side, with eyes looking towards the top of the head, this will enhance the stretch of the muscle.
This whole procedure should be performed bilaterally several times in each of the three head positions. Scalene stretches, with all their variable positions, clearly also influence many of the anterior neck structures.
Dr. Alex Jimenez offers an additional assessment and treatment of the hip flexors as a part of a referenced clinical application of neuromuscular techniques by Leon Chaitow and Judith Walker DeLany. The scope of our information is limited to chiropractic and spinal injuries and conditions. To discuss the subject matter, please feel free to ask Dr. Jimenez or contact us at 915-850-0900 .
By Dr. Alex Jimenez

Additional Topics: Wellness
Overall health and wellness are essential towards maintaining the proper mental and physical balance in the body. From eating a balanced nutrition as well as exercising and participating in physical activities, to sleeping a healthy amount of time on a regular basis, following the best health and wellness tips can ultimately help maintain overall well-being. Eating plenty of fruits and vegetables can go a long way towards helping people become healthy.

WELLNESS TOPIC: EXTRA EXTRA: Managing Workplace Stress